Bacteria ferry nanoparticles into cells for early diagnosis, treatment
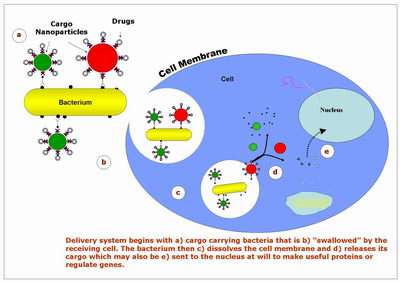
Researchers at Purdue University have shown that common bacteria can deliver a valuable cargo of "smart nanoparticles" into a cell to precisely position sensors, drugs or DNA for the early diagnosis and treatment of various diseases.
The approach represents a potential way to overcome hurdles in delivering cargo to the interiors of cells, where they could be used as an alterative technology for gene therapy, said Rashid Bashir, a researcher at Purdue's Birck Nanotechnology Center.
The researchers attached nanoparticles to the outside of bacteria and linked DNA to the nanoparticles. Then the nanoparticle-laden bacteria transported the DNA to the nuclei of cells, causing the cells to produce a fluorescent protein that glowed green. The same method could be used to deliver drugs, genes or other cargo into cells.
"The released cargo is designed to be transported to different locations in the cells to carry out disease detection and treatment simultaneously," said Bashir, a professor in the Weldon School of Biomedical Engineering and the School of Electrical and Computer Engineering. "Because the bacteria and nanoparticle material can be selected from many choices, this is a delivery system that can be tailored to the characteristics of the receiving cells. It can deliver diagnostic or therapeutic cargo effectively for a wide range of needs."
Harmless strains of bacteria could be used as vehicles, harnessing bacteria's natural ability to penetrate cells and their nuclei, Bashir said.
"For gene therapy, a big obstacle has been finding ways to transport the therapeutic DNA molecule through the nuclear membrane and into the nucleus," he said. "Only when it is in the nucleus can the DNA produce proteins that perform specific functions and correct genetic disease conditions."
When the cargo-carrying bacteria attach to the recipient cell they are engulfed by its outer membrane, forming "vesicles," or tiny spheres that are drawn into the cell's interior. Once inside the cell, the bacteria dissolve the vesicle membrane and release the cargo.
The method might be used to take images of diseased tissues by inserting a cargo of fluorescent molecules into tumors that are ordinarily too small to be detected, said Demir Akin, a research assistant professor of biomedical engineering who specializes in nanomedicine.
"These bacteria can potentially deliver specific molecules into a variety of cells," said Akin, the first author of a research paper appearing online this week in the journal Nature Nanotechnology.
Experiments were carried out in cultures of human cancer cells, including intestinal, oral, liver, ovarian and breast cancer cells. The researchers also tested their method on live mice and showed how the technique could be used to deliver specific genes to various organs, including the liver and kidneys.
"The cells in the organs receiving the bacteria with nanoparticles made the intended therapeutic proteins and emitted a light similar to a firefly's glow," Akin said.
Certain bacteria are naturally programmed to dissolve vesicle membranes, a critical step to delivering the cargo. The nanoparticles are referred to as "smart" because they release their cargo at precisely the right moment after entering the cell.
"At the same time that the bacteria are breaking up this vesicle membrane, the cargo dislodges from the bacteria, which are both crucial steps in delivering this cargo," Akin said.
The nanoparticles, which range in size from 40 to 200 nanometers - or billionths of a meter - are attached to the bacteria with "linker molecules."
"The use of commercially available polystyrene nanoparticles makes this delivery system much simpler to implement than previous alternatives," Bashir said.
This new delivery system also is more efficient than other experimental techniques using viruses and bacteria.
"With other techniques, you can usually incorporate only one copy of your gene cargo to each bacterium or virus particle," Akin said.
In the new approach, bacteria can carry hundreds of nanoparticles, each of which can in turn carry hundreds of drug molecules, depending on the size of the nanoparticles.
The approach also could make it possible to insert relatively large structures, such as sensors and hollow filaments called carbon nanotubes, into the interiors of cells.
The sensors could make it possible to monitor activities inside a single cell for the early detection of cancer and other diseases and to monitor the progress of disease and response to drug therapy. The carbon nanotubes could be delivered into diseased cells and then exposed to light, causing them to heat up and kill only those diseased cells, Akin said.
The Nature Nanotechnology paper was authored by Akin; Jennifer Sturgis and Kathy Ragheb, both on the research staff at the Bindley Bioscience Center; Debby Sherman, an electron microscopist in the Department of Biology; Kristin Burkholder, a doctoral student in the Department of Food Science; J. Paul Robinson, a professor in the Weldon School of Biomedical Engineering and the School of Veterinary Medicine; Arun K. Bhunia, a professor of food microbiology in the Department of Food Science; Sulma Mohammed, an assistant professor of cancer biology in the School of Veterinary Medicine; and Bashir. A print version will appear in July.
Source: Purdue University





















