Watching chemistry in motion: Chemical environments mapped using molecular vibrations

Scientists have long known that a molecule's behavior depends on its environment. Taking advantage of this phenomenon, a group of researchers at the University of Chicago developed a new technique to map microscopic environments using the vibrations of molecules.
"It's a special new advance that will be broadly useful in studies of molecular and materials phenomena," said Andrei Tokmakoff, the Henry G. Gale Distinguished Service Professor in Chemistry at UChicago. He and two of his associates report their new technique in a paper published online in the journal Optics Express.
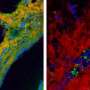
The new technique builds on ultrafast two-dimensional infrared spectroscopy, which emerged approximately 15 years ago as a method to probe molecular vibrations. When a laser pulse strikes a molecule, parts of its energy is transferred into the vibrations of the molecule. The ability of each single molecule to get rid of this excess energy, or relax, depends on the neighbors' ability to accept such energy. Thus molecules in different environments will relax at different rates, which are then used to determine the environment of individual molecules. Combining two-dimensional spectroscopy with a microscope enabled the researchers to directly visualize the microscopic variations in chemical environments.
"It's a new, hybrid technique that combines the spatial resolution of microscopy with the molecular information of infrared spectroscopy," said Carlos R. Baiz, a postdoctoral fellow and the article's lead author. The technique offers data on vibrational dynamics that traditional microscopy lacks, while adding spatial information that infrared spectroscopy alone can't provide.
"The new technique lends itself to multiple applications," said Denise Schach, a postdoctoral fellow in chemistry and co-author of the Optics Express article. "We aim to observe the protein folding process, which is the basis of biological function, inside a single cell." In the future, the new technique might especially benefit research in cellular biology and biomedicine.
Mapping vibrational frequencies
Two-dimensional IR spectroscopy can measure molecular dynamics at the femtosecond (quadrillionth of a second) timescale, which is the vibrational frequency of a chemical bond. The method is used to correlate different vibrational frequencies of a molecule, in order to learn about its structure as well as its chemical environment. Combined with microscopy, the method offers a spatial resolution of 20 microns, about the size of a human skin cell.
"Consider a system of coupled springs: you can pluck one spring and see the energy transfer from this one oscillator to all the other springs in the system," Baiz explained. "It's the same effect with molecules. The laser excites one vibration which then relaxes into other nearby vibrations on the same molecule or its neighbors, and where the vibrational energy ends up tells us about the structure and environment of the molecule."
Multiple factors contributed to the success of Tokmakoff's team, which conducted preliminary experiments for two years at MIT, that enabled the group to plot the best way to develop the new method. Once Tokmakoff joined the UChicago faculty in 2013, his startup funds financed the purchase of the sophisticated and expensive equipment that his team needed to implement the plan.
"The facilities are excellent here" said Baiz, referring to Tokmakoff's laboratory space in the Gordon Center for Integrative Science, which is equipped with stringent temperature and humidity controls, the most technologically advanced optical components, and a brand new microscope.
Also important was the purchase of a new pulse shaper that enabled the researchers to modulate individual laser pulses in a way that traditional optics cannot do, and developing a new way of collecting data that involved a different geometric alignment of the laser beams.
More information: "Ultrafast 2D IR Microscopy," by Carlos R. Baiz, Denise Schach, and Andrei Tokmakoff, Optics Express, Vol. 22, Issue 15, pp. 18724-18735, 2014. dx.doi.org/10.1364/OE.22.018724
Journal information: Optics Express
Provided by University of Chicago