Catalysts caught in the act undergo radical rearrangements during reactions
Chemists who develop catalysts are always trying to improve catalytic efficiency or create novel reaction pathways, but they're doing so largely in the dark. The atomic-scale structure and chemical properties of catalysts remain surprisingly mysterious, despite the critical roles that catalysts play in a variety of industrial and environmental applications. Heterogeneous catalysts—which differ in phase from their substrates—are used, for example, to convert toxic nitric oxide from automotive emissions to less harmful gases.
Hoping to shed some light on how catalysts behave, and connect these behaviors to catalysts' activity and selectivity, researchers working at the U.S. Department of Energy Office of Science's Advanced Photon Source teased out structural and chemical information about a single layer of vanadium oxide, a catalyst, supported on the surface of a titanium oxide crystal. The data revealed that vanadium oxide undergoes a dramatic and reversible change in both chemical states and structure during a redox reaction cycle, providing unprecedented insight into how vanadium oxide catalyzes reactions. This research will make it possible for scientists to improve catalysts by strategically altering their structures.
Supported vanadium oxide is currently under investigation as a catalyst for the oxidative dehydration of ethanol, propane, and cyclohexane. For example, low-density arrangements of supported vanadium oxide have shown greater catalytic efficiency than supported vanadium oxide at higher densities or bulk vanadium oxide. In this study, researchers from Northwestern University, the Massachusetts Institute of Technology, and Argonne National Laboratory examined the oxidative dehydration of cyclohexane by monolayer vanadium oxide on a titanium oxide scaffold as a model system.
To make the sample, the researchers atomically engineered a layer of vanadium oxide on the surface of a single rutile (10 mm x 10 mm x 1 mm) titanium oxide crystal. The researchers created vanadium atomic density maps relative to the rutile substrate lattice using x-ray standing wave (XSW) in situ analysis at the DuPont-Northwestern-Dow Collaborative Access Team (DND-CAT) 5-ID-B,C,D beamline at the Argonne Advanced Photon Source.
The key to XSW is that it uses the x-rays generated by the APS beamline in two ways: single-crystal x-ray diffraction and x-ray fluorescence. The x-ray diffraction provides the basic structural outline, while the fluorescence distinguishes between vanadium and titanium.
To run the in situ XSW experiments, the researchers placed the sample in the path of the x-ray beam inside a beryllium dome. The dome, which is "transparent" to x-rays, is used to create the environment for vanadium to catalyze chemical reactions, supplying the proper temperature, pressure, and reaction gases. To generate a three-dimensional image, the researchers rotated the crystal with a 5-circle diffractometer. They gathered structural data on the catalyst in three states: oxidized by oxygen, reduced by cyclohexane, and reoxidized by oxygen. This strategy can effectively track the behaviors of catalysts during a chemical reaction.
In addition to structural information, the researchers wanted to assess the chemical oxidation state of the catalyst under the same conditions as studied by XSW. For this, the researchers used a low-energy x-ray source provided by the Keck-II facility at Northwestern University.
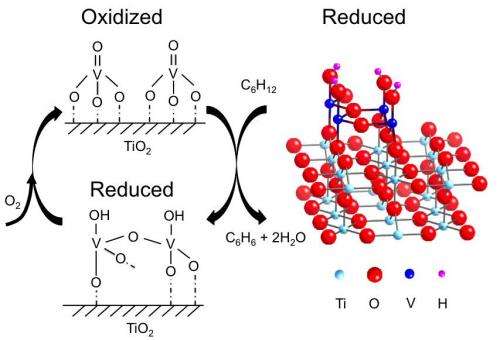
X-ray photoelectron spectroscopy (XPS) detected the oxidation state of vanadium and any bound oxygen atoms under oxidized and reduced conditions. By combining the XPS and XSW data, the researchers developed an atomic-scale model for the reduced catalyst in a 4+ oxidation state (right in the figure). Its structure strongly resembles that of the titanium oxide support, which was expected, the researchers say, because titanium also has a 4+ oxidation state.
Things got more interesting when it came to the analysis of the oxidized state. This structure was uncorrelated to that of the underlying titanium oxide, indicating that the catalyst significantly changes upon oxidation. The disharmony between the oxidized vanadium oxide and titanium oxide support made it impossible for the researchers to develop a structural model for the oxidized state.
However, using the information from XPS data as well as from infrared spectroscopy, they figured out that oxidized vanadium is in a 5+ oxidation state, with three single bonds to oxygen and one double bond (figure, top left). The oxidized and reoxidized states looked the same, suggesting that the reaction is reversible. In future research, the researchers plan to nail down the oxidized structure by combining structural and oxidation state information.
More information: Zhenxing Feng, Junling Lu, Hao Feng, Peter C. Stair, Jeffrey W. Elam, and Michael J. Bedzyk, "Catalysts Transform While Molecules React: An Atomic-Scale View," J. Phys. Chem. Lett. 4, 285 (2013). DOI: 10.1021/jz301859k
Provided by Argonne National Laboratory