Researchers obtain monodispersed metal nanoparticles with tunable high density
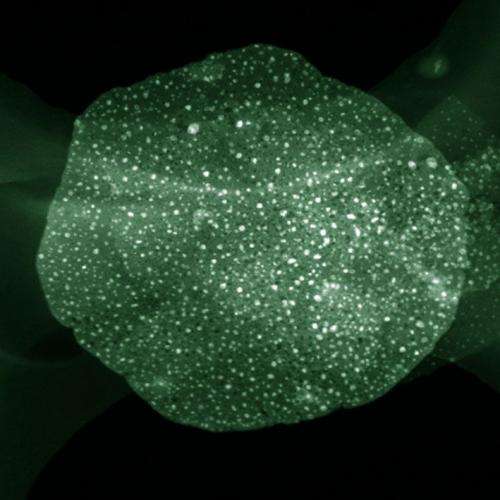
Monodispersed metal nanoparticles (NPs) obtained at Tsinghua University had tunable high density ranging from 1013 to 1016 m-2 and excellent thermal stability at a very high temperature of 900 C, according to new research.
The metal NP catalysts have been widely employed in large-volume catalytic applications such as the Haber–Bosch process for implementation of nitrogen and hydrogen, isomerization in hydrocarbon cracking for gasoline octane improvement, hydrogenation for manufacture of edible fats and oils, selective oxidation for hydrocarbon derivatives, automobile exhaust conversion for environmental protection, as well as hydrohalogenation for organic synthesis.
As catalytic reactions occur only when the reactants are adsorbed onto a working metal NP surface, a higher catalytic activity can be achieved by increasing the surface area of the active phase of the catalysts by reducing the size of the corresponding catalytic particles. Consequently, dispersing the metal NPs with tunable size, high density, and good thermal stability is a great challenge.
The team included researchers from Tsinghua University at Beijing, China; Institute of Metal Research, Chinese Academy of Sciences, at Shenyang, China; Fritz Haber Institute of the Max Planck Society at Berlin, Germany; and Technical University of Denmark, at Roskilde, Denmark. They have published their study on using an atomic metal-dispersed precursor of layered double hydroxides (LDHs) for high density, monodisperse metal NPs in Issue 6, Volume 2 of Journal of Materials Chemistry A published on Feb 14.
'LDH is a class of synthetic two-dimensional nanostructured anionic clays consisting of positively charged layers and charge-balancing interlayer anions. Most metals, such as Fe, Co, Ni, and Cu, can be well dispersed in the framework of LDHs at an atomic scale with controllable components,' said Fei Wei from Tsinghua University. In fact, LDHs are among the most important catalytic materials as a multifunctional catalyst and/or a precursor for metal-supported catalysts.
'We tried to understand the evolution of ultra-dispersed embedded metal NPs from LDHs by both in situ and ex situ methods,' Qiang Zhang from Tsinghua University told phys.org. 'The Fe/Mg/Al containing LDHs were selected as the model sample for Fe NP formation. This process was investigated by in situ X-ray diffraction together with thermogravimetric analysis, temperature programmed reduction, and high resolution transmission electron microscopy.'
They found that large quantities of mesopores induced by the Kirkendall effect were formed on the as-calcined layered double oxides (LDOs). However, the flake morphology was well preserved at a very high temperature of 900oC. Dang Sheng Su from Fritz Haber Institute of the Max Planck Society mentioned 'The O atoms bonded with Fe3+ cations were easily extracted at a temperature higher than 750oC, which greatly increased the mobility of Fe. The coalescence of the reduced Fe atoms into large NPs enhanced the Kirkendall effect, leading to the formation of monodispersed embedded Fe NPs on the porous LDO flakes.'
Under the guidance of the in situ study, the areal density of the as-prepared 1-20 nm Fe NPs on LDOs can be well mediated ranging from 0.039 to 2.1 × 1015 m-2 by adjusting the Fe content in the LDH precursors, the reduction time and temperature. Carbon nanotube (CNT) growth was selected as the probe reaction to evaluate the catalytic performance of the monodispersed metal NP catalysts. Zhang explained, 'growth of CNTs can be directly correlated with the metal NP catalysts and the growth history was recorded. For instance, when the density of Fe NPs is low, only entangled CNTs were obtained; aligned CNTs will be available when the catalyst density is large.' A phase diagram of CNT with CNT architectures from entangled CNTs, aligned CNTs, and double helical CNTs grown on flat/flake substrates with different catalyst densities and sizes can be found in this publication.
As Prof. Zhang, Su, and Wei point out, this work provides not only new insights on the chemical evolution of monodisperse NPs from an atomic metal-dispersed precursor, but also a general route to obtain tunable NPs as heterogeneous catalysts for chemical and material production. In the future, the researchers hope to precisely control the ultra-uniform metal NPs formation through the chemical interaction of metal and support during the facile reduction process. 'More precise control can be achieved by mediating the Kirkendall diffusion and chemical reduction,' Qiang Zhang said, 'An ultimate goal is to achieve customized metal NPs easily and accurately for nanocatalysis and energy conversion.'
More information: Tian GL, Zhao MQ, Zhang BS, Zhang Q, Zhang W, Huang JQ, Chen TC, Qian WZ, Su DS, Wei F. "Monodisperse embedded nanoparticles derived from an atomic metal-dispersed precursor of layered double hydroxide for architectured carbon nanotube formation. Journal of Materials Chemistry A 2014, 2(6): 1686-1696. pubs.rsc.org/en/content/articl … g/2014/ta/c3ta14380e
Journal information: Journal of Materials Chemistry A
Provided by Tsinghua University


















