Enzymatic hydrolysis of cellulose in aqueous ionic liquids

Total enzymatic hydrolysis of the polysaccharides in lignocellulosic biomass to monosaccharides is currently a focus research area. The monosaccharides obtained from lignocellulose hydrolysis can be used for the production of platform chemicals and biofuels, most notably ethanol. One major challenge in the commercialization of lignocellulosic ethanol production is the recalcitrance of lignocellulosics towards enzymatic hydrolysis, necessitating efficient pretreatment of the lignocellulosic feedstock.
Certain ionic liquids (ILs, salts with melting points below 100 °C) dissolve cellulose and even lignocellulosic biomass and are as such interesting candidates for pretreatment technology. However, cellulose-dissolving ILs have been found to severely inactivate the hydrolytic enzymes (cellulases) employed in cellulose hydrolysis. This work focuses on elucidating how certain ILs affect the action of cellulases in cellulose hydrolysis. The main emphasis was on the action of purified monocomponent Trichoderma reesei cellulases, but some commercial cellulase preparations were also studied in IL matrices.
Hydrolysis experiments were made in solutions containing up to 90% of the two cellulose-dissolving ILs 1-ethyl-3-methylimidazolium acetate ([EMIM]AcO) and 1,3-dimethylimidazolium dimethylphosphate ([DMIM]DMP). The presence of increasing amounts of IL led to decreasing yields of solubilised saccharides in enzymatic hydrolysis. [EMIM]AcO was generally more harmful for cellulase action than [DMIM]DMP. Pure [EMIM]AcO completely inactivated T. reesei endoglucanase in 4 h in residual activity measurements, whereas pure [DMIM]DMP supported considerable cellulase activity for at least three days. The cellulase compatibility of several novel classes of cellulose-dissolving ILs were studied in hydrolysis, but these ILs were found to be at least as harmful for cellulase action as the studied imidazolium-based ILs. T. reesei endoglucanases were unable to reduce the molecular weight of microcrystalline cellulose (MCC) in buffer or in any aqueous matrix containing IL, except in 90% (v/v) [DMIM]DMP in which the MCC was partially dissolved.
The studied ILs were found to have very detrimental effects on saccharide analytics. A capillary electrophoresis (CE) method was developed for the analysis of mono- and oligosaccharides in matrices containing ILs.
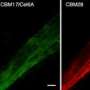
The cellulase binding to MCC in solutions with [DMIM]DMP and [EMIM]AcO was studied with radiolabeled T. reesei Cel5A (endoglucanase II)and Cel7A (cellobiohydrolase I) and their respective core domains. Cel7A was able to bind to MCC with its core domain, whereas it was shown that Cel5A was very dependent on its CBM for efficient substrate binding. [EMIM]AcO interfered more with cellulase substrate binding than [DMIM]DMP. The binding ability of the T. reesei carbohydrate-binding modules (CBMs) was very IL sensitive.
More information: Read the paper here: www.vtt.fi/inf/pdf/science/2014/S52.pdf
Provided by VTT Technical Research Centre of Finland