What makes superalloys super: Hierarchical microstructure of a superalloy
Materials in high-performance turbines have to withstand not only powerful mechanical forces, they also have to maintain their chemical and mechanical properties almost up to their melting points. For this reason, turbine manufacturers have employed special nickel-based high-performance alloys for decades. New work from Helmholtz-Zentrum Berlin für Materialien and Energie now shows in detail how new phases in a nickel-based alloy form and evolve, providing clues to how high-performance alloys could be improved.
Doctoral student Florian Vogel and Dr. Nelia Wanderka from the HZB Institute of Applied Materials have elegantly combined two methods to accomplish this: transmission electron microscopy (TEM) and atom probe tomography (APT), which they carried out in collaboration with colleagues from the University of Münster.
They were interested in a phase separation phenomenon that has been known for around 50 years, but could neither be precisely observed nor understood until now: The microstructure of nickel-based alloys changes under controlled ageing or heat treatment and in the classical two-phase microstructure new phases are initially formed. Wanderka and Vogel were able to precisely observe the phase separation process on the atomic scale for the first time.
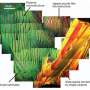
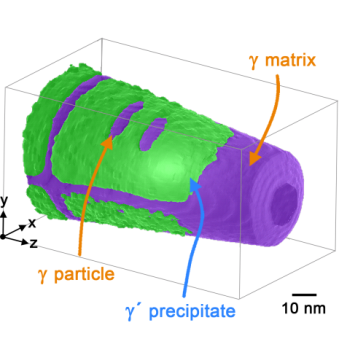
To do so, they simulated the ageing process of the alloy by heat treating it for different periods. They documented how the microstructure changed during the ageing of the alloy using micrographs from the transmission electron microscope. Whereas the classical two-phase microstructure consists of cuboid γ' precipitates embedded in a so called γ-matrix, during heat treatment, spherical γ particles initially form in the γ' precipitates of the alloy, then further coalesce into plates that finally split the γ' precipitates. The thermo-mechanical properties of these types of alloys depend largely on the stability of this γ/γ´ microstructure.
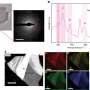
In order to determine the atomic constituents of the individual phases, but primarily to learn about the formation and make-up of the poorly understood γ particle, Vogel and Wanderka investigated the aged samples using atom probe tomography at the University of Münster. They succeeded in reconstructing the atomic lattice of the samples layer by layer and determining the composition of all phases, so that they could explain the chemical evolution of the γ particles.

"Until now, it was assumed that splitting of the γ' precipitates refines the microstructure during ageing, which would be beneficial for the alloy's stability under thermo-mechanical load. We were able to show that this is not correct. The microstructure indeed changes considerably, but it is not improved by the splitting however. We were actually able to correlate the best mechanical properties with the presence of spherical or plate-like γ particles and not with the later stages after splitting of the γ' precipitates has taken place", explains Florian Vogel. Nelia Wanderka adds: "If we want to improve the stability of the microstructure and thus the thermomechanical properties of the alloy, we need to be sure that the γ' precipitates are not split by the γ particles, but instead remain intact through appropriate heat treatment and composition of the alloy. Atom probe tomography helps us in understanding the role of the alloying elements in the formation and growth of the γ particles. From this, we can learn how to influence these processes."
More information: The work was published 20 Dec 2013 in the distinguished journal Nature Communications. www.nature.com/ncomms/2013/131 … full/ncomms3955.html
Journal information: Nature Communications
Provided by Helmholtz Association of German Research Centres