Tracking micropollutants in Lake Geneva

Antibiotics, urban pesticides, and other contaminants accumulate where wastewater is released into Lake Geneva. Using computer simulations, EPFL researchers have shown that the risk they pose is highest during summer and that they degrade most efficiently during the winter.
Once taken, antibiotics do not simply disappear. Released from the body by urine, some escape water treatment plants unscathed. Traces of antibiotics and a variety of other molecular contaminants such as pesticides and pharmaceutical drugs are consistently measured in Vidy Bay, Lake Geneva, above the site where processed wastewater is released. The impact of micropollutants is receiving ever more attention from the scientific community, but it is still unclear how long they persist and to what extent they pose a threat to organisms. In an article published in late July, researchers at EPFL present findings on the dilution of the micropollutants by lake currents. Their results showed that the pollutants are most persistent during summer, several meters under water. In the winter, they rise to the lake surface and are most effectively degraded by sunlight
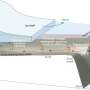
Every second, between one and three tons of treated wastewater from the city of Lausanne gush out into Lake Geneva from a pipe that ends 700 meters offshore and 30 meters below the lake's surface. Lake currents disperse and dilute this water in the same way that wind disperses the smoke from a chimney, with a persistent underwater pollutant plume emanating from the discharge pipe.
Florence Bonvin from EPFL's Environmental Chemistry Lab has been studying the dynamics of this plume and the pollutants it contains. "We consistently detect a range of micropollutants – antibiotics, anti-inflammatory and anti-epileptic drugs and urban pesticides in particular in the vicinity of the wastewater treatment plant outfall," she says. Although the concentrations are very small, she says, they are frequently well above threshold concentrations that are considered to have no effect on organisms.
Driven by lake currents
Actually getting on a boat and sampling for micropollutants is time-intensive, expensive, and only provides a partial snapshot of the situation at any given time. A computer model gives a more complete image. "We would have to sample hundreds of sites simultaneously – which is basically impossible – to come close to getting as much detail as we can using a computer model," says Bonvin. This way, she can study two simultaneously occurring processes in great detail: dilution of the micropollutants by mixing with the surrounding lake water, and their degradation by sunlight.
The dynamics of the pollutant plume are mostly driven by lake currents, which vary with the season and the weather. Summers see the lake's surface waters warm up, while the depths remain cool. The interface between warm and cold water – the thermocline – acts as a cap, trapping currents and the pollutant plume several meters under water. But as the lake's surface waters cool down during the winter, the thermocline disappears and the plume rises to the surface. With increased exposure to surface winds and sunlight, pollutants are diluted and degraded much more effectively.
The computer model lets Bonvin quantify the ecotoxicological threat posed by 24 different micropollutants as they enter the lake, disperse into the surrounding water, and eventually degrade. To study two different lake circulation patterns driven by the most prevalent winds, such as the bise, both with and without the summer thermocline, Bonvin used results from a 3D lake circulation model developed by Amir Razmi from EPFL's Ecological Engineering Laboratory, which reproduced lake currents measured during an experimental campaign carried out in Vidy Bay. The degradation of the pollutants by the sun were based on measurements she performed herself in the lab.
"Using our simulation results, we can draw a map of where the pollutants are depending on the season, the winds, and the cloud cover," says Bonvin. And they offer a way to evaluate the potential risk they pose. "The concentrations of each pollutant are very low, but since we don't know how living organisms respond to being exposed to these pollutants over long time periods, this isn't an issue we can simply ignore."
Bonvin previously showed that even antibiotics that have been processed and inactivated in our body and then released into the lake could be revived when exposed to sunlight, returning to their original, active form. This adds another source of micropollutants to the picture. Her findings, which add an additional twist to this story, were published earlier this year in Environmental Science & Technology.
More information: pubs.acs.org/doi/abs/10.1021/es401294c
Journal information: Environmental Science & Technology
Provided by Ecole Polytechnique Federale de Lausanne