A microbe's fountain of youth
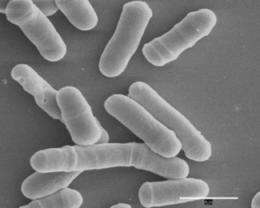
The yeast S.pombe is one of the best-studied microbes in the world. First isolated from East African millet beer over a century ago, it's been used as a model organism in molecular and cell biology for the past sixty years. And yet scientists have now just uncovered what may be its most striking feature—that under favorable conditions, it doesn't grow old.
Most single-cell organisms age. When they divide in half, one daughter cell typically receives older, defective material, while the other acquires newer components.
But S.pombe employs a different strategy. In a study published recently in Current Biology, a team of researchers has shown that, when unstressed, the yeast divides by splitting the damage equally. In a sense both daughters are "rejuvenated," since they're left with less damage than the mother cell.
"We have shown, for the first time, that there is a microbe immune to aging," says lead researcher Iva Toli?-Nørrelykke, of the Max-Planck Institute for Molecular Cell Biology and Genetics in Germany.
"I think it's the simplest strategy," adds Miguel Coelho, first author of the paper and now post-doctoral researcher at Harvard University. "If the cell grows and divides fast enough, the damage is diluted with each division. And provided that the total amount of damage does not overcome a certain death threshold, the cells can in theory divide indefinitely."
It's been known since its early discovery that S.pombe reproduces symmetrically, by dividing into two cells of the same shape and size. For this reason some researchers had speculated that there may not be a mother cell, that both daughter cells could be equally young. The yeast S. cerevisiae, for comparison, divides asymmetrically by producing a larger aging mother cell, and a smaller, fully rejuvenated, daughter cell. However, some studies had shown that even cells that divide symmetrically could still split the damage unequally inside, and thus age.
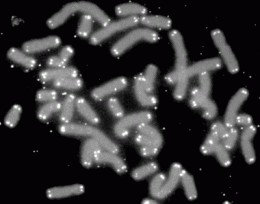
"But these studies were done by looking at single cells," says Coelho. "And to show aging you have to follow them in context of the whole colony over many generations."
So that's what the team did. "We followed the cells up to 50 generations, and measured their division times," says Toli?-Nørrelykke. Aging cells take longer and longer to divide. "The results were astonishing: the cells did not slow down their division even after tens of generations, suggesting that they were not aging," she says.
Then they looked inside the yeast cells by tagging protein aggregates, the most conserved form of age-related damage, with a fluorescent marker. They saw that, under favorable conditions, the protein aggregates were diluted down and divided equally between the two daughters. Under stressed conditions, however, the cells did split the damage unevenly, producing an aging mother and a younger daughter cell.
In certain types of mammalian cells, aging is linked to the length of telomeres—segments of DNA that get shorter and shorter with each division. But S.pombe cells, like stem and cancer cells, have an enzyme called telomerase which regenerates the length of the telomeres when they get too short. That's why researchers focus on age-related damage such as protein aggregates—not on telomere length—to study aging in yeast.
The results of this study, however, do not mean that the unstressed cells are immortal. "Some cells were still dying," explains Toli?-Nørrelykke, "but it did not result from aging—there was no increase in division time before death. We call these events "catastrophic death." The causes are likely random mutations and other random events."
There's also an important caveat: because the researchers didn't see any sign of aging doesn't mean it isn't occurring. "Proving the non-existence of something is very difficult," says Eric Stewart, a microbiologist at Northeastern University who was not involved in the study. "Their findings are definitely interesting. And they did try to look at as many aging factors as they could. But you never know if you have the right marker."
S.pombe may not be the only microbe immune to aging, says Coelho. "I don't think it's something unique to fission yeast," he explains. "Some studies have shown that other organisms who divide symmetrically, like E.coli, do age. But it may depend on the growth conditions."
Next, the study researchers plan to compare the strategies used by different microbes species. Coelho has gone on to study how the damage is processed and segregated inside the cell. These results will be published early next year. S.pombe could also be used as a model organism for other non-aging cell types—such as germ cells, stem cells, and cancer cells.
Even if the findings don't provide new insights into the origins of life on Earth, one of astrobiology's main goal, it does offer an important lesson. We have just uncovered a striking new fact about a microbe that had been studied extensively for decades. It's a good reminder that there's still much we don't know about life, and that some of the strategies which evolve can be unexpected. We just never know when the next discovery will emerge.
Journal information: Current Biology
Provided by Astrobio.net