Researchers develop new system to better study behavior, cell function
A team of researchers led by Charles D. Nichols, PhD, Associate Professor of Pharmacology at LSU Health Sciences Center New Orleans, has successfully translated a new technology to better study behaviors and cellular function to fruit flies. This powerful genetic tool – Designer Receptors Exclusively Activated by Designer Drugs – allows scientists to selectively, rapidly, reversibly, and dose-dependently remotely control behaviors and physiological processes in the fly. The fruit fly shares a significant degree of similarity to humans and can be used to model a number of human diseases including Alzheimer's, Parkinson's, cancer, obesity, diabetes, heart disease, epilepsy, ALS, mental illness, and more. The research, published on September 5, 2013, is available online in the journal, Cell Reports.
"Significant advantages the fly offers as a model are the advanced genetic tools available for manipulating gene expression, like the ability to selectively express genes in any defined cell or tissue, in combination with its prolific and rapid reproduction cycle and ease of growth," notes Charles D. Nichols, PhD, Associate Professor of Pharmacology at LSU Health Sciences Center New Orleans.
In order to study behaviors, a common method is to manipulate the activity state of neurons and observe the effects. By and large, current methods are essentially switches to turn the neuron on or off and can produce dramatic changes in neuronal activity that can manifest in significant behavioral changes. One disadvantage with these switch-like approaches is that they can mask more subtle functions of neuronal circuits in regulating behaviors.
Another is that many require expensive specialized equipment like light sources and fiber optics to manipulate neuron function. Designer Receptors Exclusively Activated by Designer Drugs, or DREADD, technology was first developed by one of the co-authors (B.L. Roth) for mammalian systems and overcomes many of the limitations of switch-based approaches.
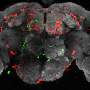
Using genetic technology, the research team developed DREADD receptors in the flies' neuronal circuits, which they selectively activated by feeding the flies a drug that only activates the DREADD receptors in their food. There were three DREADD flavors – two activating and one inhibitory. Depending on which DREADD was expressed where, distinct neural circuits could either be activated or suppressed. Because the researchers activated DREADDs with drugs in the food, the degree of activation could be precisely controlled by just changing the amount of drug in the food.
"We have successfully translated this technology to the fly and in this paper report reversible and dose responsive control of many behaviors including sensory perception, learning and memory, and courtship," says Dr. Nichols. "Our new pharmacogenetic approach fills an important and unmet need in our ability to understand fly behaviors by allowing us for the first time to conveniently examine behaviors in only partially activated neurons to uncover more subtle roles for particular neurons and circuits in behaviors." A better understanding of fly behaviors will translate to a better understanding of human biology and diseases.
A unique feature of this DREADD system not found in other methods is that it is not limited to the control of neurons – activity levels of certain enzymes in almost every tissue type can be controlled to probe mechanisms of basic cellular function. The researchers demonstrated this ability by stopping the fly heart with activation of the inhibiting DREADD expressed in the heart, and then restarting the heart simply by washing away the drug.
Journal information: Cell Reports
Provided by Louisiana State University