A greener, more sustainable source of ingredients for widely used plastics

A new process can convert a wide variety of vegetable and animal fats and oils—ranging from lard to waste cooking oil—into a key ingredient for making plastics that currently comes from petroleum, scientists say. Their report on the first-of-its-kind process appears in the journal ACS Sustainable Chemistry & Engineering.
Douglas Neckers and Maria Muro-Small explain that many of the plastics found in hundreds of everyday products begin with a group of chemical raw materials termed olefins that come from petroleum. They include ethylene, propylene and butadiene, which are building blocks for familiar plastics like polyethylene, polyester, polyvinyl chloride and polystyrene. The scientists sought a more sustainable alternative source of olefins.
Their report describes use of "UV-C" light—used in sanitizing wands to kill bacteria and viruses around the house—to change lard, tallow, olive oil, canola oil and waste canola cooking oil into olefins. Neckers and Muro-Small say that this is the first report on use of this photochemical process to make olefins.
More information: "A Green Route to Petroleum Feedstocks: Photochemistry of Fats and Oils" ACS Sustainable Chem. Eng., Article ASAP DOI: 10.1021/sc400135y
Abstract
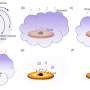
We demonstrate that it is possible to generate long chain olefins from different fat sources, i.e., animal fat, vegetable oils/fats, and waste cooking oil. Our results show that, independent of the source of fat, irradiation using UV–C light produces 1-tetradecene, 1-hexadecene, 1,7-hexadecadiene, and 1,7,10-hexadecatriene. Fats undergo transesterification with primary alcohols rendering the corresponding esters. These, following irradiation, produce the olefins described, in addition to lower molecular weight esters.
Provided by American Chemical Society