Stem cell clues uncovered
Proper tissue function and regeneration is supported by stem cells, which reside in so-called niches. New work from Carnegie's Yixian Zheng and Haiyang Chen identifies an important component for regulating stem cell niches, with impacts on tissue building and function. The results could have implications for disease research. It is published by Cell Stem Cell.
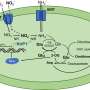
Lamins are proteins that the major structural component of the material that lines the inside of a cell's nucleus. Lamins have diverse functions, including suppressing gene expression. It has been difficult to understand how mutations in lamins cause diseases in specific tissues and organs, such as skeletal muscles, heart muscle, and fat.
A group of human diseases called laminopathies, which include premature aging, are caused by defects in proteins called lamins. Zheng and her team, which included Xin Chen of Johns Hopkins University, decided to examine whether lamins would link stem cell niche function to healthy tissue building and maintenance.
To understand the tissue-specific effects of lamin mutations, the team focused on fruit fly testis, one of the best-studied stem cell niche systems. In the fruit fly testis, biochemical cross-signaling between the different types of cells that make up the niche environment ensures proper maintenance and differentiation of the testis system's stem cells.
Using an advanced array of techniques available in fruit fly studies, the team demonstrated that lamins were a necessary component of supporting niche organization, which in turn regulates proper proliferation and differentiation of germline stem cells in fruit fly testis.
"These results could have implications for the role of lamins in other types of stem cell niches," Zheng said. "These findings could contribute to the study of diseases caused by lamina-based tissue degeneration. For example, different lamin mutations could disrupt the organization of different niches in the body, which then leads to degeneration in tissues."
Journal information: Cell Stem Cell
Provided by Carnegie Institution for Science