Jumping crystals: Kinematic analysis of light-induced jumping crystals
Live beings are not the only things that can move around – it turns out that small crystals can also rotate or even jump. Scientists from United Arab Emirates and Russia have now systematically examined crystals that move when irradiated by light. In the journal Angewandte Chemie they present the first quantitative kinematic analysis of this phenomenon, which they have termed the photosalient effect.
When irradiated with UV light, micrometer- to millimeter-sized crystals of the cobalt coordination complex [Co(NH3)5(NO2)]Cl(NO3) cover distances over thousands of times larger than themselves. Why do they do this? The nitrite ligand (NO2) is normally bound to the central cobalt ion through its nitrogen atom. This bond is broken by the irradiation and the ligand rotates a little to use one of its oxygen atoms to bind to the cobalt instead. This isomerization produces strain in the crystal. The strain is dissipated through movement and fracturing. The crystals jump and may even explode.
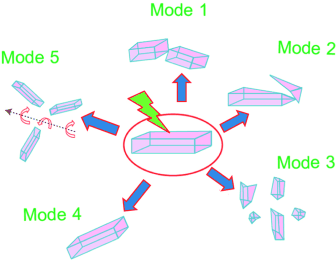
A team led by Pan?e Naumov (New York University Abu Dhabi) and Elena V. Boldyreva (Russian Academy of Sciences and Novosibirsk State University) has now systematically analyzed this effect with a microscope-mounted, high-speed camera. The scientists distinguished the following phenomena: 1) splitting of the crystal into two roughly equally sized pieces, 2) splintering off of small pieces, 3) explosion of the crystal, 4) displacement without visible splintering or lifting off the surface, and 5) rolling or jumping. These result in some complex movement sequences of the crystals and their splinters.
The distance covered depends on the duration and intensity of the irradiation. The crystals only jump after a certain period of latency during which stress builds up. When it reaches a threshold, the stress is released all at once. Smaller crystals start flipping sooner than larger ones. Interestingly, the intensity of the irradiation also determines the type of effect. Intermediate intensities primarily cause rolling and jumping, higher intensities cause more splitting off of debris. The highest intensities primarily cause the crystals to split into two equal pieces.
The scientists are convinced that the effects result from a cooperative mechanism. The rotation of individual ligands causes small intramolecular perturbations that are spread and amplified over the network of hydrogen bonds connecting the ions within the crystal lattice. This network acts like a spring that is wound by the irradiation and relaxes through the movement or splitting of the crystal. The rigidity of the springs was confirmed in precise single-crystal diffraction experiments where the sample was exposed to high pressure.
The conversion of light energy to mechanical movement could be useful for the design of materials that imitate the movement of animals, or dynamic technical components in devices like nanomachines.
More information: Dynamic Single Crystals: Kinematic Analysis of Photoinduced Crystal Jumping, Angewandte Chemie International Edition, dx.doi.org/10.1002/anie.201303757
Journal information: Angewandte Chemie , Angewandte Chemie International Edition
Provided by Wiley