Two proteins that control nuclear DNA distribution also regulate development by altering gene expression
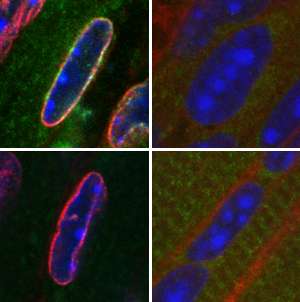
Cell biologists believe that gene expression in eukaryotic cells is partly controlled by the uneven distribution of DNA in the nucleus. Colin Stewart and Audrey Wang at the A*STAR Institute of Medical Biology, Singapore, and their international co-workers, have identified two proteins that control this distribution of DNA. Their findings have important implications for disease and cellular development.
Heterochromatin is DNA that is tightly packed to prevent expression of its genes, and it is mainly tethered to the inside of the nucleus wall, also called the 'nuclear envelope'. Euchromatin is DNA that is loosely packed and ready for gene expression, and it is in the center of the nucleus.
Stewart and Wang discovered that DNA distribution depends on two proteins found in the nuclear envelope: lamin B receptor (LBR) and lamin A/C (lamA/C). They discovered that neither of these proteins is expressed in a type of photoreceptor cell that naturally has a reversed distribution of heterochromatin and euchromatin. When the researchers removed both LBR and lamA/C from normal mouse cells, they observed the same reversal, proving that the absence of these proteins is responsible for this DNA distribution.
According to Stewart, the involvement of LBR and lamA/C has important consequences. "Mutations in the lamin proteins cause a range of inherited diseases, including muscular dystrophy, enlargement of the heart, premature aging and diseases affecting skeletal development and fat production," he explains. "These [conditions] are rare, but studying them may provide insights into more pressing diseases, such as cardiovascular disease, obesity and aging."
Looking at the roles of LBR and lamA/C in more detail, Stewart and Wang also found that the proteins are expressed at different times during embryonic development; LBR is expressed initially, but it is replaced over time by lamA/C. In muscle cells, the two proteins also had opposite effects on the expression of muscle-specific genes: LBR silenced the genes, whereas lamA/C switched them on. The combination of differential expression and gene activation allows the two proteins to orchestrate cellular development.
The team's work answers some questions about the mechanisms and importance of uneven DNA distribution. Stewart notes, however, that there are now many more questions to investigate. "Is the disruption to heterochromatin a cause or consequence of diseases? What does this effect on chromatin structure mean for the stability of the genome? Does it increase susceptibility to other diseases, such as cancer?" he asks. "These are avenues that we will now pursue."
More information: Solovei, I., Wang, A. S., Thanisch, K., Schmidt, C. S., Krebs, S. et al. LBR and lamin A/C sequentially tether peripheral heterochromatin and inversely regulate differentiation. Cell 152, 584–598 (2013). www.cell.com/abstract/S0092-8674%2813%2900012-3
Journal information: Cell
















