Papaya-clay combo could cut cost of water purification in developing countries

An inexpensive new material made of clay and papaya seeds removes harmful metals from water and could lower the cost of providing clean water to millions of people in the developing world, scientists are reporting. Their study on this "hybrid clay" appears in the journal ACS Sustainable Chemistry & Engineering.
Emmanuel Unuabonah and colleagues explain that almost 1 billion people in developing countries lack access to reliable supplies of clean water for drinking, cooking and other key uses. One health problem resulting from that shortage involves exposure to heavy metals such as lead, cadmium and mercury, released from industrial sources into the water. Technology exists for removing those metals from drinking water, but often is too costly in developing countries. So these scientists looked for a more affordable and sustainable water treatment adsorbent.
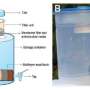
They turned to two materials readily available in some developing countries. One was kaolinite clay, used to make ceramics, paint, paper and other products. The other: seeds of the Carica papaya fruit. Both had been used separately in water purification in the past, but until now, they had not been combined in what the scientists term "hybrid clay." Their documentation of the clay's effectiveness established that the material "has a strong potential for replacing commercial activated carbon in treatment of wastewater in the developing world."
More information: "Hybrid Clay: A New Highly Efficient Adsorbent for Water Treatment" ACS Sustainable Chem. Eng., Article ASAP DOI: 10.1021/sc400051y
Abstract
New hybrid clay adsorbent based on kaolinite clay and Carica papaya seeds with improved cation exchange capacity (CEC), rate of heavy metal ion uptake, and adsorption capacity for heavy metal ions were prepared. The CEC of the new material is ca. 75 meq/100 g in spite of the unexpectedly low surface area (≈9 m2/g). Accordingly, the average particle size of the hybrid clay adsorbent decreased from over 200 to 100 μm. The hybrid clay adsorbent is a highly efficient adsorbent for heavy metals. With an initial metal concentration of 1 mg/L, the hybrid clay adsorbent reduces the Cd2+, Ni2+, and Pb2+ concentration in aqueous solution to ≤4, ≤7, and ≤20 μg/L, respectively, from the first minute to over 300 min using a fixed bed containing 2 g of adsorbent and a flow rate of ≈7 mL/min. These values are (with the exception of Pb2+) in line with the WHO permissible limits for heavy metal ions. In a cocktail solution of Cd2+, and Ni2+, the hybrid clay shows a reduced rate of uptake but an increased adsorption capacity. The CEC data suggest that the adsorption of Pb2+, Cd2+, and Ni2+ on the hybrid clay adsorbent is essentially due to ion exchange. This hybrid clay adsorbent is prepared from materials that are abundant and by a simple means that is sustainable, easily recovered from aqueous solution, nonbiodegradable (unlike numerous biosorbent), and easily regenerated and is a highly efficient alternative to activated carbon for water treatment.
Provided by American Chemical Society