When benzene's bonds break: Hydrogen release depends on bond scission, not absorption

(Phys.org) —In adding steam to benzene, C6H6, to generate hydrogen, the step that determines the reaction's speed is not the benzene's absorption onto the catalyst, but rather the first benzene bond that breaks, according to scientists at Pacific Northwest National Laboratory. The team further explored increasing the speed of the hydrogen-producing reaction by evaluating rhodium and iridium catalysts on a magnesium aluminum spinel support. Through experimental and computational studies, they found that small rhodium particles had a higher turnover efficiency than either larger rhodium particles or iridium. This research graced the cover of ACS Catalysis.
"Our research focuses on the development of reforming catalysts that can be applied to a host of technologies, such as biomass gasification, pyrolysis, and natural gas—and will further the understanding for how catalysts function at the molecular level," said Robert Dagle, who led this experimental research at PNNL.
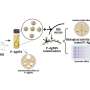
Pacific Northwest National Laboratory is helping drive the shift from petroleum to bio-based fuels by developing the scientific and engineering foundations for converting biomass to biofuels that are infrastructure ready. In the syngas upgrading to hydrocarbon fuels pathway, biomass feedstocks are gasified to produce a syngas (H2/CO), which is used as a feedstock for hydrocarbon biofuel production. By applying advanced scientific instruments and computational chemistry, the researchers determined the first steps in benzene, a model tar surrogate, and water's conversion to carbon monoxide and hydrogen.
"These initial steps are very important. With these steps, you learn how to stop problems before they start," said Dr. Vanda Glezakou, a PNNL theorist who worked on this 3-year study.
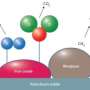
The team is developing alternative, atom-efficient catalysts that have the potential significantly improve tar reforming performance over the traditionally used nickel-based hydrocarbon-reforming catalysts. The research team examined the benzene steam-reforming reaction with rhodium or iridium catalytic particles on a magnesium-based spinel, MgAl2O4. With help from using a Titan 80-300 aberration-corrected transmission electron microscope at EMSL and other instruments, they determined the reaction does not depend on the amount of steam added, which is indicative of benzene's absorption on the catalyst. This suggests that the first benzene decomposition step, not the adsorption, is the rate-determining step on either rhodium or iridium catalysts.
However, the team found that the reaction changed depending on the catalyst used. "We started asking questions about what the particle size is doing in terms of helping the catalytic route," said Dr. Roger Rousseau, the theory group leader.
Using density functional theory calculations, the team examined how the support controlled the size of the catalyst and how other factors affected the reaction. The calculations showed that smaller rhodium particles lowered the energy barriers involved with breaking carbon-carbon and carbon-hydrogen bonds. For example, the activation barrier for the carbon-carbon bonds dropped from 1.60 eV on the larger rhodium particles to 1.34 on the smaller particles. The team saw a similar drop in the energy barrier between the larger and smaller iridium particles, but did not see a corresponding drop in the carbon-hydrogen bond scission.
"After the first bond break, it is kind of complicated," said Dr. Donghai Mei, the lead computational theorist for this project and first author of the scientific article in ACS Catalysis. For example, on the rhodium catalysts, the team's calculations showed that the carbon-carbon and carbon-hydrogen bond scissions are competitive, regardless of the rhodium particle size. After the first bond breaks, the rhodium-catalyzed reaction favors carbon-hydrogen breaks.
This fundamental understanding of breaking bonds is critical not only in developing efficient tar-reforming catalysts, but also in other applications. "We can use this knowledge for a wide range of fuel processing technologies," said Rousseau. "It is really about renewable fuels."
The team is continuing to examine the effect of the spinel support on other catalytic reaction. They are specifically examining the role of the spinels in determining the optimum size of the catalyst's particles and the resulting chemistry. In addition, the team is extending this research into understanding the fundamentals of reforming the organic species found in the aqueous phases of bio-oils that are derived from a variety of biomass liquefaction routes.
More information: Mei, D. et al. 2013. Comparative Investigation of Benzene Steam Reforming over Spinel Supported Rh and Ir Catalysts, ACS Catalysis 3, 1133-1143. pubs.acs.org/doi/abs/10.1021/cs4000427
Journal information: ACS Catalysis
Provided by Pacific Northwest National Laboratory