Rapid threat assessment could mitigate danger from chemical and biological warfare
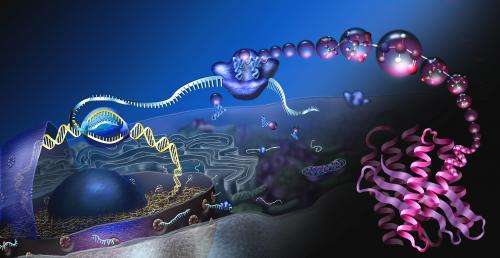
For more than fifty years, researchers have been studying exactly how aspirin affects the human body. Despite thousands of publications on the topic, our understanding is still incomplete.
Meanwhile, novel chemical and biological weapons have historically been mass produced within a year of discovery. Using current methods and technologies, researchers would require decades of study to gain a robust understanding of how new threat agents exert effects on human biological systems.
That capability gap leaves U.S. forces vulnerable, so DARPA's new five-year program, Rapid Threat Assessment (RTA), sets an aggressive new goal for researchers: develop methods and technologies that can, within 30 days of exposure, map the complete molecular mechanism through which a threat agent alters biochemical processes in human cells. The developed technologies must identify the cellular components and mechanistic events that take place over a range of times, from the milliseconds immediately following exposure to the threat agent, to the days over which alterations in gene and protein expression might occur. The molecular mechanism must also account for molecular translocations and interactions that cross the cell membrane, cytoplasm and nucleus.
Details of the program are available at: go.usa.gov/TGfm . DARPA will host a Proposers' Day on May 21; registration information is available at: go.usa.gov/TGtT .
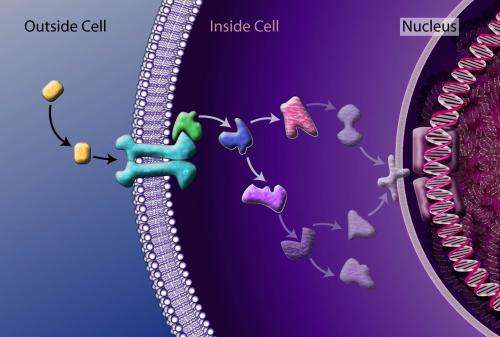
"Understanding the molecular mechanism of a given threat agent would provide researchers the framework with which to develop medical countermeasures and mitigate threats," said Barry Pallotta, DARPA program manager. "If the RTA program is successful, potential adversaries would have to reassess the cost-benefit analysis of using chemical or biological weapons against U.S. forces that have credible medical defenses."
Threat agents, drugs, chemicals and biologics interfere with normal cell function by interacting with one or more molecules associated with the cell membrane, cytoplasm or nucleus. Since a human cell may contain up to 30,000 different molecules functioning together in complex, dynamic networks, the molecular mechanism of a given threat agent might involve hundreds of molecules and interactions. The technologies sought by RTA would identify the molecular targets of threat agents and the complex interactions that follow.
"Introducing a threat agent into a cell sets off a chain of interactions that propagate throughout the cell much like the pattern of ripples that result from throwing a pebble into a pond," said Pallotta. "Unfortunately, current research tends to be highly specialized, examining effects on very specific proteins or lipids and so on, which is why a drug like morphine is still being studied almost 200 years after its introduction. For this reason, DARPA is demanding a comprehensive approach that identifies all of the affected components and interactions at once against a background of inherent complexity."
To help navigate this complexity, program performers will have to discover the series of events in the molecular mechanism at time intervals from initial introduction of a threat agent to a period of several days of exposure. This could be accomplished by freezing cells at various points in time to capture interactions as they unfold. This approach will help researchers discover the actual sequence of molecular events initiated by the threat agent.
While providing a framework for the development of medical countermeasures to chemical and biological weapons, successful RTA technologies would also be readily applicable to drug development and treatments for disease. In both cases, detailed knowledge of molecular mechanism is one of the ingredients that enable new drugs to win approval or diseases to be treated.
DARPA hopes to pair technologies developed during the RTA program with its Microphysiological Systems program, which is building "human-on-a-chip" technology. By introducing threat agents or proposed countermeasures to the human-on-a-chip system, researchers could observe which human cell types are affected, and use RTA technologies to elucidate the specific molecular mechanisms in the affected cells.
Provided by DARPA