For superionic material, smaller is better
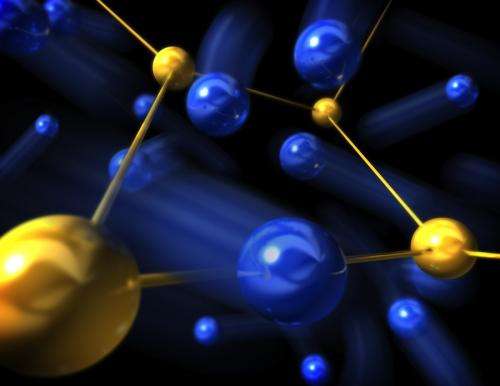
(Phys.org)—A material that could enable faster memory chips and more efficient batteries can switch between high and low ionic conductivity states much faster than previously thought, SLAC and Stanford researchers have determined. The key is to use extremely small chunks of it.
"Our result is a step toward using this material, copper sulfide, in low-cost solid-state electrical batteries," said the leader of the research team, Aaron Lindenberg, of the Stanford Institute for Materials and Energy Sciences and the Stanford PULSE Institute. The institutes are run jointly by SLAC and Stanford.
"For the first time, we've seen the atomic-scale details of exactly how these nanoscale materials transform, or switch, from a state that is poorly conducting to one that is highly conducting," he said. "And what we've learned gives us confidence about our ability to tune its structure and properties to be useful in new technologies."
Lindenberg's team reported its results last month in Nature Communications.
Copper sulfide is a "superionic" solid material: When heated above a critical temperature it suddenly conducts current, in the form of ions moving through the material, much more readily than at lower temperatures. This transition into a superionic phase has fascinated researchers since the 1830s, when it was first described by the famous British scientist Michael Faraday.
With recent advances in nanoscience, scientists have learned that smaller pieces of superionic material have lower critical temperatures and may also switch states much faster. For instance, a centimeter-sized piece of copper sulfide becomes an excellent conductor when heated above 103 degrees Centigrade, but its fastest recorded switching times occur on a scale of millionths of a second.
In the most recent advance, Lindenberg's team found that much tinier nanodisks of copper sulfide, just 10 nanometers (10 billionths of a meter) across, switched at lower temperatures – about 70 degrees C. – and can be controlled by light to switch in only 20 trillionths of a second, a million times faster than previously observed.
Superionic materials typically have two constituents – in this case, copper and sulfur –whose structures are intertwined. The new report shows in exquisite detail what happens inside the material when the critical temperature is reached, and indicates a connection between the time it takes for switching to occur and the motion of the ions through the superionic material when it's at equilibrium.
"This paper presents the first experimental measurements capturing a superionic nanocrystal as it transforms and the associated changes within the transition state itself," Lindenberg said. "We were surprised how quickly the crystal form and volume changed."
Now that they know the details of the superionic transformation, Lindenberg says his team has ideas for synthesizing new classes of superionic materials that are stable at room temperature, as well as for exploring the way ions are transported through the material and how the associated transitions from one phase to another take place.
Such an improvement could enable copper sulfide to be used as a key element in new classes of resistive switching devices and as a safe, efficient solid-state replacement for the liquid electrolyte that transports ions between the electrodes of a rechargeable battery.
More information: www.nature.com/ncomms/journal/ … full/ncomms2385.html
Journal information: Nature Communications
Provided by SLAC National Accelerator Laboratory