Computational study of human hair provides insights into structure of its poorly understood outer surface
Human hair is a complex, multi-layered material, the composition of which is only partly established. Hair fibers are sheathed in a thin protective coating called the epicuticle, but despite its industrial importance—the epicuticle is the first surface with which hair products interact—the exact structure of this layer is unknown. Now, a theoretical model of epicuticle structure developed at the A*STAR Institute of High Performance Computing (IHPC) has revealed the likely composition and properties of hair's outer surface. The model, developed by Daniel Cheong and his co-workers at IHPC, is already helping to resolve apparent discrepancies over epicuticle structure.
Previous experimental research has shown that hair's outermost surface consists of a thin monolayer of fatty acid molecules called 18-MEA, which stand on end like the bristles on a brush. According to Cheong, one study suggested that these molecules attach to the surface around 1 nanometer apart. "Although this distance is frequently cited, it has never been corroborated, as it is very difficult to measure this value experimentally," says Cheong.
To examine the 18-MEA separation distance further, Cheong and his co-workers constructed simplified computational models of the hair surface. They then looked for the separation distance that gave the most energetically stable structure. "Surprisingly, our simulation results indicated that the separation distance between the fatty acids should be around 0.5–0.65 nanometers," says Cheong.
One possible explanation for this apparent disagreement with earlier work could be that bound 18-MEA molecules are indeed spaced 1 nanometer apart; but extra, unbound lipids may pack the space in between to generate the more stable structure, Cheong suggests.
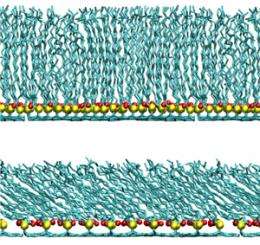
This theory could also resolve apparently conflicting results regarding experimental measurements of the fatty acid layer's thickness, which have ranged from 1.3 nanometers to 2.6 nanometers. The team's model shows that the more tightly the fatty acids are packed, the more upright they stand, which makes the epicuticle appear thicker (see image). Cheong suggests that, in studies where this layer was found to be only 1.3 nanometers thick, the free lipids may have been lost, partly collapsing the fatty acid structure. Tellingly, his model predicts that for fatty acids spaced 1 nanometer apart, the layer would appear 1.3 nanometers deep.
"With this simple model, we can also study the interactions between small molecules and the hair surface," Cheong explains. "This would be important in understanding how potential active ingredients in hair products will behave at the hair surface."
More information: Cheong, D. W., Lim, F. C. H. & Zhang, L. Insights into the structure of covalently bound fatty acid monolayers on a simplified model of the hair epicuticle from molecular dynamics simulations. Langmuir 28, 13008–13017 (2012). pubs.acs.org/doi/abs/10.1021/la302161x
Journal information: Langmuir