New technology could improve treatment for diabetes, other disorders

(Phys.org) -- For people suffering from diabetes and other hormonal disorders, staying healthy means staying vigilant; effective treatment requires periodic and precise doses of drugs throughout the day.
The constant upkeep can make for an inconvenient and stressful way of life, but managing these disorders could become easier thanks to drug-delivery technology being developed by Zhengdong Cheng, associate professor in the Artie McFerrin Department of Chemical Engineering at Texas A&M University.
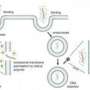
Cheng’s work, which is detailed in the scientific journal Drug Delivery Letters, is exploring a new method for manufacturing the tiny particles that once inside the bloodstream deliver drug treatments to targeted areas of the body. Unlike previous models, Cheng’s drug delivery particles could potentially administer periodic doses of the drug they contain throughout the day – something that has historically proven difficult for drug-delivery technology but if achieved could eliminate the need for a patient to self-administer multiple times, he says.
Typically, drug delivery vehicles are limited by a single mode of release, meaning a drug is released into the body in a slow but continuous manner or all at once, Cheng notes. For some treatments, that’s ok, but such a delivery method won’t work for everything – think of how a diabetic has to administer insulin at specific times throughout the day.
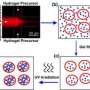
For these instances, a periodic release is required, and to achieve this Cheng has taken the construction of these drug-delivery particles in a new direction, pairing something called an ion exchanger with a pH-sensitive hydrogel, the material that forms his drug-delivery particle.
With the help of the ion exchanger, the drug-delivery particle undergoes some complex reactions that result in the particle being made to expand and contract in an oscillating manner, Cheng explains. With every expansion, the “gate” of the particle essentially opens, allowing the drug to pass into the affected area of the body. With every contraction, the “gate” closes, and the drug cannot be released into the body, Cheng says.
Think of the ion exchanger as a sort of converter at the molecular level, Cheng says. It’s made from microscopic crystals embedded in the drug-delivery particle, and it helps change the pH levels inside the particle, he explains.
Specifically, the ion exchanger exchanges sodium ions to produce protons and form molecular compounds, Cheng notes. Initially, sodium ions diffuse into the hydrogel and trigger the ion-exchange reaction. As part of the reaction, protons are generated and the pH level inside the hydrogel decreases, which causes contraction.
Later when the pH level is low enough, the ion exchange reaction works in an opposite manner to restore equilibrium, he explains. As part of this process, hydroxide ions diffuse into the hydrogel, neutralizing the protons and producing water. This neutralizes the acid, and the hydrogel absorbs water and swells, Cheng says. The swelling and shrinking cycle can repeat several times before stopping, depending on the availability of sodium and hydroxide ions and the ion exchange process.
Although there is still work to be done before Cheng’s mechanism becomes a viable drug-delivery vehicle, preliminary results are promising, he says, noting that the particles have demonstrated the ability to deliver a payload with a system. He plans to continue researching the model to further explore methods of controlling the oscillating effects in different environments as well as with different payloads.
Provided by Texas A&M University