Researchers discover cellular system for detecting and responding to poisons and pathogens
Two Massachusetts General Hospital (MGH)-based research teams, along with a group from the University of California at San Diego, have discovered that animals have a previously unknown system for detecting and responding to pathogens and toxins. In three papers published in the journals Cell and Cell Host & Microbe, the investigators describe finding evidence that disruptions to the core functions of animal cells trigger immune and detoxification responses, along with behavioral changes.
"Viewing many diseases through the prism of this newly discovered system will eventually allow a reinterpretation of disorders from several branches of medicine as aberrant responses to toxins and bacteria," says Gary Ruvkun, PhD, of the MGH Department of Molecular Biology, senior author of a paper in the April 13 issue of Cell. "While these initial studies are in the C. elegans roundworm, many of the regulatory factors that we have identified are also present in humans."
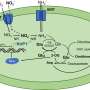
The Cell paper from the Ruvkun lab describes experiments by research fellow Justine Melo, PhD, revealing that inactivation by RNA interference of cellular components involved in core cellular functions – including translation of messenger RNAs into proteins on ribosomes or energy production in mitochondria – not only halted growth and reproduction in C. elegans but also induced the animals to move away from the E. coli bacteria they usually prefer to consume. Several of these deactivated components are known to be targets of chemical or protein toxins produced by bacteria and fungi, and Melo and Ruvkun showed that C. elegans exhibited the same aversive behavior when exposed to benign E. coli supplemented with any of several natural chemical toxins.
Additional experiments by Melo and Ruvkun revealed that inactivation of these core cellular components induced expression of genes known to be involved in the innate immune system's response against specific toxins and pathogens even when no toxins or pathogens were actually present. The researchers theorize that the observed behavioral response of C. elegans is similar to the way other animals avoid eating when they feel ill, whether or not food is the source of illness. They note that a fundamental cellular surveillance system that responds broadly to toxin-produced disruption of essential activities rather than to the presence of the toxin itself could protect against pathogens not previously encountered, and that directly monitoring these core components would allow early detection of and response to unknown toxins.
One of the reports appearing in Cell Host & Microbe from a team led by Frederick Ausubel, PhD, of MGH Molecular Biology reported similar results. That study found that consumption by C. elegans of E. coli induced to express a protein-synthesis-inhibiting toxin produced by the bacterial pathogen P. aeruginosa activated innate immune gene pathways that are also induced by protein-synthesis-inhibiting antibiotics produced by different pathogens. These pathways were not activated by an inactive version of the P. aeruginosa toxin that did not affect protein synthesis, indicating that it was the disruption of that core cellular activity and not the presence of the toxin itself that produced the immune response. The accompanying Cell Host & Microbe study from the UC San Diego team led by Emily Troemel, PhD, found that this same P. aeruginosa toxin also activates synthesis of a key immune regulator, directly stimulating the response against the toxin.
"It has been predicted for quite a while that animal immune systems would respond to the effects of bacterial toxins, but there has not been a lot of experimental support for this hypothesis," says Ausubel. "The experiments described in our Cell Host & Microbe paper, however, show very clearly that C. elegans can detect a disruption of protein synthesis that leads to a strong immune response, irrespective of whether they have been challenged with a pathogen. We are now testing whether the mammalian immune system responds the same way."
Ruvkun notes that these genetic pathways for responding to pathogens and their toxins will probably be important in many human diseases. Individual variations in responses to pathogen-produced toxins could explain the runaway inflammation and organ failure of sepsis and toxic shock, and drugs developed to target this pathway could help combat those potentially life-threatening responses. Activation of these pathways also may underlie nausea – which may be the human version of C. elegans aversion to toxin-laced food – a major problem plaguing both drug development and the use of current therapies such as chemotherapy drugs, making suppression of these responses a significant goal.
"This genetic analysis of how toxins are detected and the signals that are generated may identify new endocrine pathways in the worm and corresponding versions in humans," says Ruvkun, a professor of Genetics at Harvard Medical School (HMS). "Further study may reveal how these endocrine signals relate to human drug response and whether the endocrine state of these aversively stimulated animals corresponds to that of humans who have been 'poisoned' with pathogen-produced toxins. Identifying the genetic components of this aversive behavior could lead us to discover the endocrinology behind feeling ill and new ways to relieve that universal response."
Provided by Massachusetts General Hospital