Biology-oriented synthesis (BIOS) - cancer drugs based on natural models
The search for new drug candidates for cancer treatment is laborious and time-consuming. Only a fraction of the promising chemical compounds identified completes the transition to the market. Herbert Waldmann and his team at the Max Planck Institute of Molecular Physiology in Dortmund are therefore working on the development of methods that enable the faster discovery and synthesis of bioactive substances. The researchers have already broken one world record: in the longest known reaction cascade to date, they synthesised centrocountin molecules that inhibit cell growth.
In order to find a new substance that may one day find its way into a new cancer drug, scientists must first detect suitable drug candidates among all of the possible molecules. Once they have discovered a promising molecule, they must synthesise it and test its effectiveness before its further development into a drug can begin – all in all a laborious and, above all, time-consuming undertaking.
In the early 1990s, researchers pinned their hopes on the assembling of as many molecules as possible with the help of synthesis robots, and then trawling through these vast substance libraries for suitable compounds with the help of automated procedures. However, the initial enthusiasm for this approach soon turned into disillusionment: very few of the newly discovered molecules displayed any effect on living cells, and almost none of them completed the process of development into a finished product.
As a result, Herbert Waldmann and his colleagues at the Max Planck Institute of Molecular Physiology in Dortmund are looking for more efficient methods. The researchers’ main concern here is to both limit the search and simplify the associated synthesis process. “There are simply far too many different compounds to search randomly on the off chance that you’ll stumble onto something,” says Herbert Waldmann. The chemical structural space that includes all possible drug-like structures contains an estimated 1062 different molecules – a number, which, written out in full, fills two thirds of a line on a closely spaced typed page. Therefore, pre-selection is the most important thing.
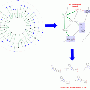
To this end, the scientists search the chemical structural space using an ingenious computer program called Scaffold Hunter, which was developed at the Max Planck Institute in Dortmund. Scaffold Hunter generates maps of a selected chemical structural space based on structural criteria and enables the researchers to navigate the sea of possible molecules and approach islands of biological activity on the computer screen. “It actually works as simply as a video game,” says Herbert Waldmann. When navigating, the Scaffold Hunter searches for structural motifs that resemble already known structures with a particular biological characteristic. Because chemically related compounds are also very likely to have similar properties, the researchers can track down promising structures in this way. They can then use these as a basis and experiment with different chemical appendages to synthesise new compounds.
But how do the researchers actually know what they should look for? “We take direction from models found in nature,” Herbert Waldmann says, explaining the principle of biology-oriented synthesis (BIOS), based on which the Dortmund scientists carry out their work. “Natural substances were selected over the course of evolution to fulfil important tasks, mostly by binding to a certain protein receptor.” Many of these substances, which arise in animals, plants or microorganisms not only affect their actual target, but also influence human cells. The plant substances morphine and digitalis, which are used for pain relief and in the treatment of heart disease, are a well-known example of this. In total, over one third of all drugs are based on natural substances; in the area of cancer therapy, the proportion is even higher.
Such natural substances and other substances closely related to them are usually complex in structure - the second biggest challenge for the chemists. In order to synthesize the substances, the scientists usually must carry out numerous individual steps in sequence, and repeatedly isolate and purify the intermediate products before they finally obtain the desired end product. Synthesis robots usually fail when faced with the complexity of this task: because they can only master comparatively simple conversions, they can only produce small molecules whose structures are not very complex. Thus, the synthesis of natural substances requires manual work.
However, as Herbert Waldmann and his colleagues have shown, elegant methods are also available for this task. Using a so-called reaction cascade, the team succeeded in synthesising active substances from the centrocountin group – complex molecules with four ring systems in the middle which intervene in cell division and may, therefore, point the way to new anti-tumour drugs. And as is so often the case in science, the discovery arose by chance, as the researchers were actually aiming to synthesise an entirely different molecule. “But the reaction did not unfold as planned and we unexpectedly set a world record,” says Herbert Waldmann. The reaction cascade included a total of twelve steps – a length that has not yet been exceeded in cascade synthesis.
To set the reaction in motion, the researchers simply provided tryptamine and formylchromone and added two catalysts. All of the conversions then proceeded completely automatically, like a domino effect in which all of the tiles fall in succession, once the first one is toppled. The entire synthesis took place in a single vessel and included nine different individual reactions, in which two catalysis mechanisms were involved. “The synthesis of such complex molecules using traditional methods takes days, if not weeks,” says Kamal Kumar, who made a significant contribution to the development of the synthesis method. With the reaction cascade, the production process was successfully completed within a maximum of 30 minutes.
Once the researchers had isolated the new compounds, they wanted to establish, via tests on cell cultures, whether the molecules would have an effect on living cells. They made an important discovery here: following treatment with the centrocountins, during the division phase, instead of two daughter cells, three or more were produced from one cell. However, they were not viable. “The effect is due to the fact that the centrocountins influence the formation of the spindle apparatus,” explains Herbert Waldmann. This spindle-shaped structure usually forms on opposite sides of the dividing cell and ensures that chromosomes are halved and pulled back into the two daughter cells.
As the scientists demonstrated, following the addition of centrocountins, the cells form not two but several attachment points for the spindle apparatus, known as the centrosomes. Because the cell no longer appears to be capable of counting its centrosomes, the researchers gave the substances the name “centrocountins”. Due to the presence of numerous centrosomes, chromosomes are unevenly divided between the daughter cells. The division cycle then comes to a halt and programmed cell death is triggered in the newly produced cells – they commit suicide so to speak.
Could this effect possibly also be used to cause tumour cells to commit suicide? This is the hope of the scientists in Dortmund. The effect of the centrocountins arises from the fact that they bind to two proteins called NPM and Crm1. Both play an important role in the formation of the spindle apparatus and are, therefore, potential target molecules for cancer treatment. “A drug that binds to both NPM and Crm1 has not existed up to now,” says Slava Ziegler, another scientist in Herbert Waldmann’s team.
The researchers do not yet know the precise mechanism by which the newly-discovered substances influence the function of the two proteins. For this reason, they are now focusing on clarifying the biochemical processes involved. When they have obtained this information, they then aim to synthesise a compound based on the centrocountins which could become a possible active agent candidate – and with a lot of luck, eventually find its way into a new cancer drug.
Provided by Max-Planck-Gesellschaft