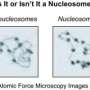
Elusive Z- DNA found on nucleosomes
New research published in BioMed Central's open access journal Cell & Bioscience is the first to show that left-handed Z-DNA, normally only found at sites where DNA is being copied, can also form on nucleosomes.
The structure of DNA which provides the blueprint for life has famously been described as a double helix. To save space inside the nucleus, DNA is tightly wound around proteins to form nucleosomes which are then further wound and compacted into chromatin, which is further compacted into chromosomes.
But this familiar image of a right handed coil (also called B-DNA) is not the only form of DNA. At sites where DNA is being copied into RNA (the messenger which is used as the instruction to make proteins) the DNA needs to unwind, and, in a process of negative supercoiling, can form a left-handed variety of the DNA double helix (Z-DNA).
It was originally thought that Z-DNA could only be formed in the presence of active RNA polymerase (the enzyme which assembles RNA). However more recently it has been discovered that SWI/SNF, a protein involved in remodeling nucleosomes and allowing RNA polymerase access to DNA, can convert certain sequences of B to Z-DNA.
The team of researchers led by Dr Keji Zhao discovered that they could convert B-DNA to Z-DNA on nucleosomes by the addition of SWI/SNF and ATP (the cell's energy source) and that the Z-nucleosome formed was a novel structure.
Dr Zhao, from the NIH, explained, "The fact that we have found Z-DNA on nucleosomes is a new step in understanding the roles of chromosome remodeling and Z-DNA in regulating gene expression. While the Z-nucleosome is likely to be a transient structure it nevertheless provides a window of opportunity for the placement of DNA binding proteins which may recruit, regulate, or block the transcription machinery and hence protein expression."
More information: SWI/SNF-mediated chromatin remodeling induces Z-DNA formation on a nucleosome Niveen Mulholland, Yan Xu, Hiroshi Sugiyama and Keji Zhao Cell & Bioscience (in press)
Provided by BioMed Central