Mystery of car battery's current solved

(PhysOrg.com) -- Chemists have solved the 150 year-old mystery of what gives the lead-acid battery, found under the bonnet of most cars, its unique ability to deliver a surge of current.
Lead-acid batteries are able to deliver the very large currents needed to start a car engine because of the exceptionally high electrical conductivity of the battery anode material, lead dioxide. However, even though this type of battery was invented in 1859, up until now the fundamental reason for the high conductivity of lead dioxide has eluded scientists.
A team of researchers from Oxford University, the University of Bath, Trinity College Dublin, and the ISIS neutron spallation source, have explained for the first time the fundamental reason for the high conductivity of lead dioxide.
A report of the research appears in this week’s Physical Review Letters.
‘The unique ability of lead acid batteries to deliver surge currents in excess of 100 amps to turn over a starter motor in an automobile depends critically on the fact that the lead dioxide which stores the chemical energy in the battery anode has a very high electrical conductivity, thus allowing large current to be drawn on demand,’ said Professor Russ Egdell of Oxford University’s Department of Chemistry, an author of the paper.
‘However the origin of conductivity in lead oxide has remained a matter of controversy. Other oxides with the same structure, such as titanium dioxide, are electrical insulators.’
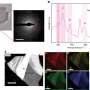
Through a combination of computational chemistry and neutron diffraction, the team has demonstrated that lead dioxide is intrinsically an insulator with a small electronic band gap, but invariably becomes electron rich due to the loss of oxygen from the lattice, causing the material to be transformed from an insulator into a metallic conductor.
The researchers believe these insights could open up new avenues for the selection of improved materials for modern battery technologies.
Professor Egdell said: ‘The work demonstrates the power of combining predictive materials modelling with state-of-the-art experimental measurements.’
Journal information: Physical Review Letters
Provided by Oxford University