New process could advance use of healthy cells or stem cells to treat disease
In a discovery that may help speed use of "cell therapy" — with normal cells or stem cells infused into the body to treat disease — scientists are reporting development of a way to deliver therapeutic human cells to diseased areas within the body using a simple magnetic effect. Their report appears in ACS' journal Langmuir.
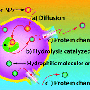
Rawil Fakhrullin and colleagues explain that cell therapy aims to replace damaged or diseased cells in the human body with normal cells or stem cells. To do so, medical personnel need a way to target these cells to diseased organs or tissues. So-called superparamagnetic iron oxide nanoparticles (SPIONs), attached to therapeutic cells, show promise. Magnetic devices could be used to move such cells to diseased areas of the body. But current ways of attaching SPIONs to therapeutic cells are difficult to use and may damage the therapeutic cells. So the researchers set out to develop a better process for attaching SPIONs to human cells.
They describe a new process for making "stabilized" SPIONs in the laboratory and successful attachment of these magnetic nanoparticles to the outside of human cells. They found that the SPIONs were not toxic to cells, and they moved in response to a magnet. "Our current results, as we believe, will inspire scientists to apply the simple and direct technique reported here in tissue engineering and cell-based therapies," say the researchers.
More information: A Direct Technique for Magnetic Functionalization of Living Human Cells, Langmuir, 2011, 27 (23), pp 14386–14393. DOI: 10.1021/la203839v
Abstract
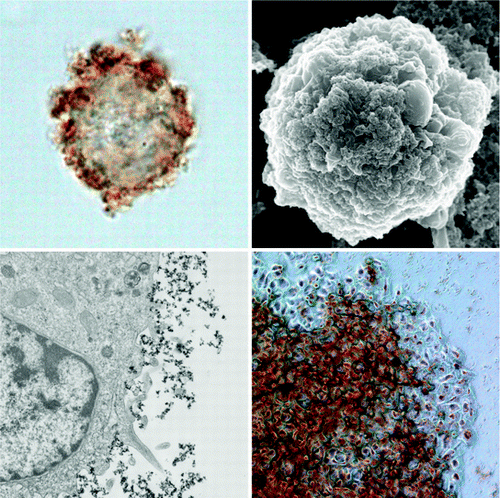
Functionalized living cells are regarded as effective tools in directed cell delivery and tissue engineering. Here we report the facile functionalization of viable isolated HeLa cells with superparamagnetic cationic nanoparticles via a single-step biocompatible process. Nanoparticles are localized on the cellular membranes and do not penetrate into the cytoplasm. The magnetically responsive cells are viable and able to colonize and grow on substrates. Magnetically facilitated microorganization of functionalized cells into viable living clusters is demonstrated. We believe that the technique described here may find a number of potential applications in cell-based therapies and in development of whole-cell biosensors.
Journal information: Langmuir
Provided by American Chemical Society