UC chemistry research looks to turn food waste into fuel

Three Department of Chemistry undergraduate researchers recently spent time at the Ho Chi Minh City (HCMC) University of Science, in Vietnam, working on ways to turn food waste into biodiesel fuel. Cody Nubel, Megan McCallister and Zachary Herrmann joined Professor Allan Pinhas and graduate researchers, Amos Doepke and Chau Phung, on a joint research project with Lawrence Pratt of Fisk University.
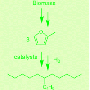
The project, “Biodiesel Fuel from Food Waste,” focuses on the potential to produce biodiesel from oil extracted from garbage. With the depletion of oil reserves, research into alternative fuels has exploded, especially in the area of renewable resources such as garbage and food waste. One particular resource, brown grease from food waste, has yet to realize its full potential, but thanks to the efforts of University of Cincinnati chemistry researchers, the project is finding ways to optimize this resource.
One of the goals of the project was to determine the amount of usable oil that can be obtained from food waste. To do this, the researchers collected food waste—by hand— from an HCMC University of Science student canteen as well as a private residence. Samples of the food waste were then either sun-dried or dried in an oven—a process Nubel describes as “extremely dirty and smelly”—and then ground up and loaded into a homemade extraction thimble bag so that oils contained in the food could be extracted through a Soxhlet extraction.
It was during the extraction process that Nubel made an important discovery. “During this step I determined a much more efficient way of pulling remnants of oil from the bag by using the suction formed from the extractor.”
McCallister, who also helped with the food waste collection, sample preparation and oil extraction, adds that after the extraction, “we converted the oil into biofuel through an acid-based reaction.”
Part of the conversion process requires that the extracted oil be degummed to further purify it. “During the degumming process we realized that layer separation is best completed through centrifuge rather than other conventional methods because it’s both higher yielding and much quicker—by about 24 hours,” says Nubel.
From there, the oil was converted to biodiesel fuel by using a solvent, which required the researchers to determine the correct amount of solvent to add per time and heat in order to yield the highest amount of biodiesel fuel.
Despite the complexity of the process—and the sometimes unseemly conditions—the results were worth the effort. “This job may have easily qualified for Mike Rowe’s show, ‘Dirty Jobs,’ but the experiment was a success and the results are awaiting publication,” says Nubel.
Herrmann also worked on the project, though in a slightly different area. He worked on taking vegetation found in Vietnam and extracting compounds from them. In a process similar to the Soxhlet extraction, Herrmann used column chromatography, which meant he also had to grind up the plants so that compounds could be extracted. “My job was to run these columns and attempt to purify one compound from thousands. We did manage to separate one compound from the plant we worked on and it was the first time that the compound had been extracted from that plant,” he says.
Not only was the research project a success, but so were the researchers themselves. Pinhas says, “What I was told in Vietnam is that the group of students from UC was the best group of students that ever participated in this program with chemists in Vietnam.”
Provided by University of Cincinnati