New class of drugs for the reversible inhibition of proteasomes
As the "recycling plant" of the cell, the proteasome regulates vitally important functions. When it is inhibited, the cell chokes on its own waste. Cancer cells, in particular, are very sensitive because they need the proteasome for their uncontrolled growth. Biochemists at the Technische Universitaet Muenchen (TUM) have now identified the lead structure of a new class of drugs that attacks the proteasome in an unusual way. New medication could be developed on the basis of this previously unknown binding mechanism. The scientists report their results in the scientific journal Angewandte Chemie.
The proteasome, a large protein complex, carries out a vitally important function in the cells of the body. Similar to a recycling plant, it decomposes unneeded proteins into short pieces and recycles them. In this way it controls a number of functions in the cell. It regulates cell growth and division, decomposes damaged proteins and also acts as a key partner of the immune system in immune defense and inflammatory reactions. Because it is involved in so many important mechanisms within the cell, the proteasome is also associated with many diseases such as cancer, mucoviscidosis and a whole series of neurodegenerative disorders such as Parkinson's or Alzheimer's disease.
Due to its significant role in the growth of cancer cells, in recent years the proteasome has taken center stage in pharmacological research as a starting point for cancer medication. When it becomes inhibited, the growth of cancer cells slows down. Bortezomib, the first drug to apply this strategy, generates revenues of over one billion US dollars per year in the meantime. It is used against multiple myeloma, a cancer disease of the bone marrow. Yet in spite of all its successes, the proteasome inhibitors currently in use often have severe disadvantages. As a result of their high reactivity they attack other proteins, too, thereby damaging not only cancer cells but also other healthy cells.
The search for alternatives conducted by a group of scientists headed by Professor Michael Groll, who holds the Chair of Biochemistry in the Department of Chemistry at the TU München, in collaboration with Professor Robert Huber, Director Emeritus at the Max-Plank Institute of Biochemistry and Dr. Stefan Hillebrand from Bayer CropScience, has now borne fruit: In a high throughput screening, the scientists examined a substance library of 200,000 potential agents in their quest to identify proteasome inhibitors – and they were successful. They identified a new structure with the so-called N-hydroxyurea motif, which reacts not only reversibly but above all specifically with the active nucleus of the proteasome. The structure inhibits the function of particular subunits of the protein complexes, which are catalytically active, and thus incapacitates the enzyme. Because of this property, the newly discovered hydroxyurea structures work more specifically than other proteasome inhibitors and are thus expected to lead to less severe adverse side effects.
The scientists were already familiar with the basic hydroxyurea structure, albeit in a completely different context. The substance in question is a derivative of the agent zileuton, which is used to treat asthma. Zileuton itself does not influence the proteasome, but its derivative, which had so far received little attention, does. "We have now found a completely new application for this previously known class of substances," explains Michael Groll. "This is of great advantage, because there are already clinical trials that give us first indications of how this class of substances behaves in the human body."
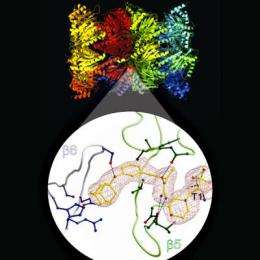
The initial structure originally discovered in the database inhibited the proteasome very specifically, but not terribly effectively. In order to modify the substance in such a way that it also works in lower concentrations – such as those required for medication – it was an important next step to understand how exactly the structure attacks the proteasome. To shed further light on this, the scientists conducted a crystal structure analysis. The outcome was that the hydroxyurea motif attacked the proteasome in a completely different manner than all other previously known inhibitors. It reacts via hitherto unknown binding pockets that may serve as starting points for the development of the new medication agents.
Starting from preliminary modeling studies, the researchers synthesized a series of different derivatives of the agent, which were then examined using X-ray crystallography and activity tests to optimize the effectiveness of the structure. The results showed that the proteasome inhibiting activity of the hydroxyurea derivatives depends on the two side chains attached to the basic structure.
Because the proteasome is contained in every cell and involved in numerous cellular functions the new inhibiting structure offers a whole range of applications, not only in the field of oncology. In the context of autoimmune diseases, an inhibition of the immunoproteasome, a derivative of the proteasome, might play an important role. In the case of autoimmune diseases, including some forms of rheumatism, the immune system attacks the body's own tissue. If the immunoproteasome is inhibited such over-reactions might be weakened. In future studies Professor Groll's team plans to improve the effectiveness of the hydroxyurea structure via experiments on cell cultures.
More information: New class of non-covalent proteasome inhibitors: the hydroxyureas, Nerea Gallastegui, Phillip Beck, Marcelino Arciniega, Robert Huber, Stefan Hillebrand and Michael Groll, Angewandte Chemie, DOI: 10.1002/anie.201106010 Link: dx.doi.org/10.1002/anie.201106010
Journal information: Angewandte Chemie
Provided by Technische Universitaet Muenchen