Chiral metal surfaces may help to manufacture pharmaceuticals
(PhysOrg.com) -- Research provides insight into novel approach which could be used in pharmaceutical drug synthesis.
New research shows how metal surfaces that lack mirror symmetry could provide a novel approach towards manufacturing pharmaceuticals.
These ‘intrinsically chiral’ metal surfaces offer potential new ways to control chiral chemistry, pointing to the intriguing possibility of using heterogeneous catalysis in drug synthesis. Such surfaces could also become the basis of new biosensor technologies.
A chiral object, such as your hand, is one that cannot be superposed on its mirror image. Chirality is fundamental in biochemistry. The building blocks of life – amino acids and sugars – are chiral molecules: their molecular structures can exist in either “left-handed” or “right-handed” forms (or “enantiomers”).
A living organism may respond differently to the two enantiomers of a chiral substance. This is crucially important in the case of pharmaceutical drugs, where the therapeutic effect is often tied strongly to just one enantiomer of the drug molecule. Controlling chirality is therefore vital in pharmaceutical synthesis.
Research into controlling chiral synthesis focuses mainly on using homogeneous catalysts, where the catalyst is in the same phase as the reactants and products, such as a liquid added to a liquid-phase reaction. However, this poses significant practical challenges in recovering the valuable catalyst material from the mixture. To avoid this problem, an attractive alternative would be heterogeneous catalysis over a solid surface – the type of catalysis used in catalytic converters in car exhaust systems, as well as in industrial Haber-Bosch synthesis of ammonia and Fischer-Tropsch synthesis of synthetic fuel, for example. The question then is how to achieve enantiomer-specific effects at a surface.
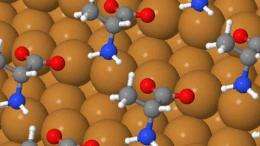
To help answer this question, scientists at the University of Cambridge have been probing the spontaneous self-organization of a simple chiral amino acid, alanine, into regular molecular arrays on copper single-crystal surfaces. Thanks to a powerful scanning tunnelling microscope, capable of resolving individual atoms and molecules, their work is revealing the various manifestations of chirality that occur, giving important clues to how they arise, and how they might be controlled and exploited.
Dr Stephen Driver, of the Department of Chemistry at the University of Cambridge, who led the experimental work, said: “We set out to investigate two distinct scenarios. In one scenario, the surface is non-chiral, so any chirality that we see can only arise from the chirality of the alanine molecule. In the other scenario, we move to a surface that is intrinsically chiral. Now the question becomes: do the two enantiomers of alanine behave differently on this chiral surface?”
On the non-chiral surface, the researchers found that alanine can self-organise into either of two patterns. In one of these, the self-organisation is driven by hydrogen bonding between the molecules, while the chiral centre has no discernable impact on the regular array. In the other structure, a network of long-range chiral boundaries punctuates the array, and the boundary chirality switches with molecular chirality.
Driver explained: “The implication is that the chiral centre is having a direct influence on the packing of two alanine neighbours at the boundary, and that the chirality of this pair propagates to the next pair and the next and so on, so that the chiral boundary is built up over a long range.”
The chiral surface is created simply by choosing a surface orientation that lies away from any of the bulk mirror symmetry planes of the metal crystal. When the researchers added alanine, they found that the surface changes its local orientation, forming nanometre-scale facets. The two enantiomers of alanine self-organise into different chiral patterns: a strong, enantiomer-specific structural effect. This “proof of principle” could potentially be exploited in chiral recognition, in chiral synthesis (forming a chiral product from non-chiral reactants), and in chiral separations.
Driver added: “It looks like alanine can shape a comfortable, chiral bonding site for itself. The copper surface has the flexibility to adapt itself to the shape of the alanine molecule, and this shape is different for the two different molecular enantiomers.”
The results imply that certain surface orientations will form stable, ordered structures with one molecular enantiomer but not the other: exactly the right conditions to promote chiral chemical effects.
Professor Sir David King, former Chief Scientific Advisor to the UK Government and current Director of the Smith School of Enterprise and the Environment at Oxford, brought together the team carrying out this research. “These results are very exciting,” said King. “Tailoring the right surface to the right molecule should lead to strong enantiospecific effects. We see a real basis here for a breakthrough technology in the pharmaceuticals sector. It’s something that pharma companies should be taking a close interest in.”
The Cambridge team’s findings are published in Topics in Catalysis.
More information: Image credit: Steve Driver
Provided by University of Cambridge