Record-breaking solar cell announced by multinational research team
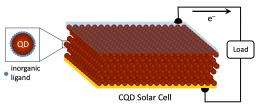
The most efficient colloidal-quantum-dot solar cell ever created will be described in a scientific paper to be published in a print edition of the journal Nature Materials by a team of scientists that includes John Asbury, assistant professor of chemistry at Penn State University. Other members of the research team are at the University of Toronto (U of T) in Canada and the King Abdullah University of Science & Technology (KAUST) in Saudi Arabia. The journal also will publish the team's achievement on its Advance Online Publication website.
"We figured out how to shrink the wrappers that encapsulate quantum dots down to the smallest imaginable size -- a mere layer of atoms," said Professor Ted Sargent at U of T, the corresponding author on the work and the holder of the Canada Research Chair in Nanotechnology. Quantum dots are nanoscale semiconductors that capture light and convert it into electrical energy. Because of their small size, the dots can be sprayed onto flexible surfaces, including plastics, enabling the production of solar cells that are less expensive than the existing silicon-based version.
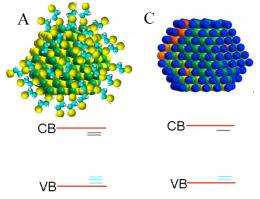
But a crucial challenge for the field has been improving their efficiency. The ideal design for greatest efficiency is one that tightly packs the quantum dots together. Until now, quantum dots have been capped with organic molecules that separate the nanoparticles by a nanometer -- making them too bulky for optimum efficiency. To solve the problem, the research team turned to inorganic ligands, sub-nanometer-sized atoms that bind to the quantum dot surfaces and take up less space.
"The inorganic ligands form the smallest possible shell that can be wrapped around quantum dots," Penn State's Asbury explains. "It is the thinness of the shell that allows the quantum dots to pack so closely that electrons can flow smoothly through the material to make photocurrent."
The colloidal quantum dots examined by Asbury and his team members yielded the highest electrical currents, and the highest overall power-conversion efficiency, ever seen in colloidal quantum dot (CQD) solar cells. These performance results were certified by an external laboratory, Newport, that is accredited by the U.S. National Renewable Energy Laboratory.

"Extensive testing has confirmed that we were able to remove charge traps -- locations where electrons get stuck -- while still packing the quantum dots closely together," Asbury said. The combination of close packing and charge-trap elimination enabled unprecedented levels of photocurrent to flow through the solar cells, thus providing record efficiency.
A technology licensing agreement has been signed by U of T and KAUST, brokered by MaRS Innovations (MI), which will enable the global commercialization of this new technology. "Through U of T's, MI's, and KAUST's partnership, we are poised to translate exciting research into tangible innovations that can be commercialized," said Sargent. "The world -- and the marketplace -- need solar innovations that break the existing compromise between performance and cost.
Provided by Pennsylvania State University

















