Building better memories with supramolecular structures that act as tiny magnets
In a step towards realizing ultrahigh-density storage devices based on individual molecules behaving as magnets, researchers in Japan have developed a candidate building block -- a supramolecular ferromagnet, which is a caged molecule with magnetic properties. The Japanese research team was led by Takuzo Aida at the RIKEN Advanced Science Institute, Wako, and Kentaro Tashiro at the National Institute for Materials Science, Tsukuba.
The researchers’ supramolecular magnet is based on a metallofullerene dubbed La@C82 -- a lanthanum ion trapped within an 82-carbon spherical cage. La@C82 has well-known paramagnetic properties: it becomes magnetized in the presence of an external magnetic field. However, like all paramagnets, La@C82 loses its magnetization once the external field is removed, rendering it useless for data storage.
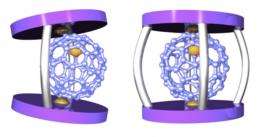
Endeavoring to overcome this loss of magnetization, Aida and his colleagues designed a molecular component that combines with La@C82 to form the supramolecular ferromagnet. They built a copper-containing structure, itself paramagnetic, to house La@C82 within an internal cavity. When mixed together, the two components self-assembled into a host–guest complex. To lock the La@C82 in place, the researchers clipped together extra arms on the host structure, converting it into a cage.
Assessing the magnetic properties of the locked and unlocked complexes, however, revealed a surprise, says Aida. Thanks to the interaction of the paramagnetic character of the two components, the unlocked caged complex did behave as a ferromagnet. However, when the researchers locked the supramolecular cage, that ferromagnetism was lost.
“It is difficult to predict the magnetic behavior of host–guest complexes,” says Aida. “We envisaged that the caged structure would give rise to a ferromagnetic property, but this was not the case.”
According to calculations run by the team, the switch in ferromagnetic behavior is all down to geometry. The La@C82 guest is a tight fit within the cavity of its host, and the unlocked cage forms an asymmetric, twisted structure. When the arms of the cage are closed, the structure is forced into a symmetrical shape for which the ferromagnetic state is no longer energetically favorable (Fig. 1).
The researchers are continuing to work with the unlocked supramolecular ferromagnet, building up arrays of the structure on a solid support—an essential step towards developing a practical memory device, says Aida. The most important point is that the cage must be properly oriented such that the resultant material retains its ferromagnetism, he says.
More information: Hajjaj, F., et al. Ferromagnetic spin coupling between endohedral metallofullerene La@C82 and a cyclodimeric copper porphyrin upon inclusion. Journal of the American Chemical Society 133, 9290–9292 (2011).
Provided by RIKEN