Researchers devise biomaterial that could be used in the detection of toxins and pollutants
(PhysOrg.com) -- In research recently published in the leading international journal PNAS, Trinity researchers exploit the potential of a biomaterial to reveal the activity of important fat metabolising enzymes. The findings show that the biomaterial could possibly be used in the future detection of toxins, explosives, pollutants, and medicines.
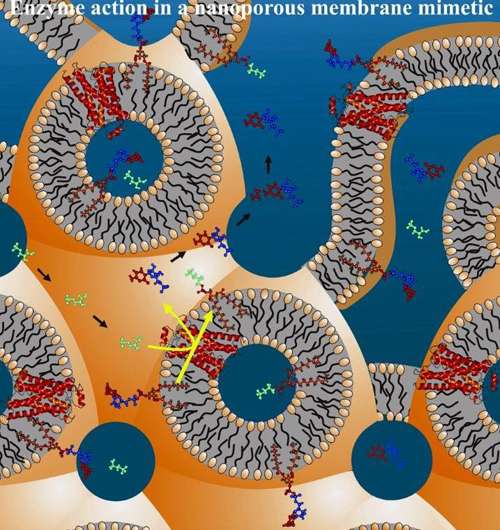
Detection devices have superior sensitivity when the sensor itself can be packaged at high density. Certain proteins that are found in the membranes of cells can act as sensors. However, the density with which cellular membranes can be packed in a sensor of a defined volume can limit the application. In this study, use was made of a particular form of matter, referred to as a liquid crystal or mesophase, that behaved as a densely packed mimic for cellular membranes.
Certain naturally occurring lipids or fats, when combined with water spontaneously form liquid crystals. One of these lipids called monoolein is a product of fat digestion. The liquid crystalline cubic phase that monoolein forms, when wet, has the lipid arranged as a bilayer just two molecules thick that is bathed on either side by water. This hydrated bilayer resembles the membrane that surrounds the cells in living organisms. The cubic phase is particularly notable as a liquid crystal in the extraordinary density with which it packages the membrane and the enormous surface area that it has. Thus, for example, a mere thimbleful of the cubic phase has enough surface area to cover a football field.
The research conducted by Trinity’s Professor of Membrane Structural and Functional Biology, Martin Caffrey and Research Associate Dr Dianfan Li in the School of Medicine and School of Biochemistry & Immunology used the cubic phase; but the cubic phase made from hydrated fat alone was useless. It needed to have a membrane protein sensor incorporated into it and the protein needed to be active. The test sensor used in the research was a membrane protein, referred to as DgkA. DgkA is an enzyme that interconverts the fatty components of natural cellular membranes. The enzyme was produced in E. coli bacteria, using recombinant DNA technology, as an inactive or dead ‘scrambled egg’ type of insoluble aggregate. ‘Life’ was breathed back into the enzyme by dissolving the aggregated protein in a soapy solution and inserting it into the membrane of the cubic phase. In this new and quite artificial environment the researchers showed that the protein had regained its original native enzyme activity and that it could behave as a model sensor.
The research sets the stage for the exploitation of this most extraordinary of biomaterials. These include its use in high density, high sensitivity biosensors for the detection of biological molecules such as hormones, proteins, carbohydrates, and lipids, as well as toxins, explosives, pollutants, and drugs.
Provided by Trinity College Dublin



















