Nature of bonding determines thermal conductivity
Optical data carriers such as DVDs, Blu-rays and CD-RWs store data in layers of so-called "phase change materials". In the future, these materials will enable the development of fast, non-volatile and energy-saving main memories. A prerequisite for this is a low thermal conductivity. Phase change materials display a surprisingly low thermal conductivity even in the crystalline state. This is described by an international research team including scientists from Julich and Aachen in the latest edition of the respected journal Advanced Functional Materials. Their findings will facilitate a targeted search for materials with the desired properties.
Phase change materials are among the favourite candidates for developing a "universal memory", which is as fast as DRAM (dynamic RAM), has high storage density, is always ready for use and does not lose data even when inadvertently turned off. The data is stored in tiny areas of different electrical resistance, which are written to by heating with the aid of electric pulses. In doing so, the atomic ordering of the material and its electrical resistance is changed.
When heated, phase change materials switch from the unordered (amorphous) to the ordered (crystalline) state, which leads to a change in their physical properties. This feature has been exploited by industry for many years in optical data carriers such as DVDs, Blu-rays and CD-RWs. By means of a laser, the atomic structure and thus the optical properties are changed in minute areas of the discs. This allows bits to be written to the disc and be read out again by a laser.
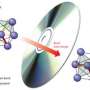
"In order to produce energy-saving and tightly packed electronic memories, it is important when the data are written to the disc that the electrical resistance is significantly changed but that the energy remains as localized as possible," explains Dr. Raphaël Hermann from the Jülich Centre for Neutron Science, who is also currently a visiting professor at the University of Liège. "Phase change materials are very well suited because they are poor conductors of heat not only in the unordered but also in the ordered state, in contrast to semiconductors, for example," adds Prof. Matthias Wuttig from RWTH Aachen University. As part of an international research team, Hermann and Wuttig are investigating the reasons for this surprising material behaviour on alloys of germanium, antimony and tellurium. With the aid of sophisticated scattering experiments at the European Synchrotron Radiation Facility (ESRF) in Grenoble, they demonstrated that the bonding conditions between the atoms in the crystalline state as well as deviations from the perfect lattice structure influence the transmission of these vibrations through the material and thus reduce its thermal conductivity.
"The starting point for our investigations was the observation by our Japanese colleagues that the amorphous material is harder than the crystalline," says Hermann. "This contradicted all assumptions, but the measured stronger bonding forces between the atoms in the amorphous state fitted the picture." The Jülich scientists investigated how the atoms in the specimens vibrate – both locally in the atomic range and also over longer distances. "In the crystalline material, we found harder vibrations for the long-range order and better conductivity for sound than in amorphous material. This is normal and is related to an increase in the order. However, we were surprised by the results for short-range vibrations in the crystal. They were softer. The short-range order in crystalline material is therefore lower than in amorphous material. This is very unusual."
On the basis of all the experimental results, the Aachen research group headed by Wuttig developed a model to explain the apparent contradictions. "Normally, the propagation of sound waves in material correlates with the thermal conductivity. However, this is not the case with phase change materials. This is due to the fact that in the crystalline state atoms experience resonance bonding – in other words, the bonding electrons are shared between several atomic pairs. In contrast, in amorphous material the atoms are covalently, that is more strongly and more locally, bonded. The crystalline material is therefore softer and the atoms vibrate more gently. In addition, there is more disorder in the local range. Both of these aspects impair the conductivity for heat carriers, which are partially of short wavelength, but not for the long-wavelength sound waves." The researchers assume that their findings will facilitate a targeted search for materials with the desired properties.
More information: Phase change materials: Vibrational softening upon crystallization and its impact on thermal properties; Matsunaga et al; Advanced Functional Materials; DOI: 10.1002/adfm.201002274
Provided by Helmholtz Association of German Research Centres