Structure formed by strep protein can trigger toxic shock
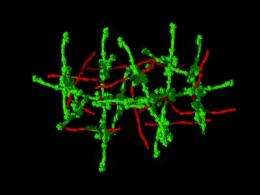
Infection with some strains of strep turn deadly when a protein found on their surface triggers a widespread inflammatory reaction.
In a report published April 7 in the journal Nature, researchers describe the precise architecture of a superstructure formed when the bacterial protein called M1 links with a host protein, fibrinogen, that is normally involved in clotting blood.
The proteins form scaffolds with M1 joints and fibrinogen struts that assemble into dense superstructures. Frontline immune cells called neutrophils mistake these thick networks for blood clots and overreact, releasing a chemical signal that can dilate vessels to the point where they leak, the team reports.
"We knew that M1 plus fibrinogen was inflammatory, but how was unknown. By determining the structure of this complex, we were able to identify the characteristics that lead to a sepsis response," said Partho Ghosh, Ph.D., professor of chemistry and biochemistry at the University of California, San Diego who studies the structure of virulence factors and led this project.
Ghosh and colleagues found that the density of the M1-fibrinogen structure was a critical characteristic. Looser structures or separate fibers formed by altered versions of M1 failed to trigger a pathological response.
"This research provides the first snapshot of the interaction between this key bacterial virulence factor and its human target at the atomic level," said Victor Nizet, M.D., professor of pediatrics and pharmacy and a co-author of the report.
Difficult to treat once they set in, the leaking blood vessels and organ failure of strep-induced toxic shock prove fatal for 30 percent of patients. Ghosh and Nizet have a long-standing collaboration aimed at designing treatments to counteract the toxic effects of strep protein.
More information: Streptococcal M1 protein constructs a pathological host fibrinogen network. DOI:10.1038/nature09967
Provided by University of California - San Diego

















