Carbonates make diamonds grow in the Earth's mantle

Diamonds that no-one ever sees can form deep in the Earth’s interior. This is due to the chemical conditions controlling the associated carbon cycle. Swiss researchers used laboratory experiments to show how this happens.
Lava that is almost as free-flowing as water and, when it cools down, as pale as limestone: the Ol Doinyo Lengai in northern Tanzania is the only active volcano in the world that produces so-called carbonatite lava. Unlike conventional lava, the majority of this lava consists not of molten silicates but of molten carbonates. Volcanoes of this kind are found along rift valleys where the continental plates gradually break apart and new oceans are formed.
Diamond from lime and iron
These carbonate melts, among other things, form when limestones enter the Earth’s mantle. This occurs as a result of subduction, when old oceanic crust plunges down below an adjoining crustal plate. By means of high-pressure experiments in the laboratory, postdoc Arno Rohrbach and ETH Zurich Professor Max Schmidt from the Institute of Geochemistry and Petrology of ETH Zurich studied how the oxidised carbon contained in the carbonates behaves in the Earth’s mantle, and in so doing they made an exciting discovery: at a depth of more than 200 kilometres, the submerged carbonates of the oceanic crust reach their melting point, which is 300 to 400 degrees lower than that of silicates. A carbonatite melt is formed and migrates from the subducted oceanic crust into the surrounding mantle, where it causes partial melting therein. However, because the Earth’s mantle is strongly chemically reducing and contains elemental metallic iron, the CO2 in the carbonatite melt reacts with the elemental iron. This causes diamonds to form.
Earlier studies have already shown that carbonates in the Earth’s mantle cause the surrounding mantle to melt at relatively lower temperatures at pressure in excess of 2.5 gigapascals, but these studies focused exclusively on the oxidised state of the carbonates. However, the ETH Zurich researchers write in their study, which was recently published on-line in Nature, that in order to understand what happens in the significantly reduced deeper mantle of the Earth it is necessary to consider the redox equilibrium between oxidised carbonates and the reduced metal-containing deeper mantle.
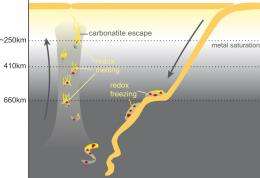
“Minerals in the Earth’s mantle such as garnet and perovskite preferentially incorporate trivalent iron during their formation”, says Max Schmidt. Part of the otherwise doubly positively charged iron is oxidised for this purpose, while another part that is half the amount of the trivalent iron is simultaneously reduced to elemental iron. Because metallic iron and carbon dioxide are incompatible, the iron metal is oxidised and the carbon dioxide is reduced to pure carbon. Thus, diamonds crystallise out of the carbonate melt at a pressure of more than 10 gigapascals and temperatures of 1400 to 1700 degrees Celsius, consequently the carbonate melt solidifies. Thereafter only triply and doubly positively charged iron together with the diamonds remain in these mantle domains in the transition zone between the upper and lower mantle at a depth between about 410 and 660 kilometres.
The carbon cycle in the Earth’s interior is more complex
However, as a result of convection in the Earth’s mantle over hundreds of millions of years, the “agglomerates” of diamonds, which cover a distance of between one and ten centimetres per year, can be transported into the lower mantle down to a depth well in excess of 2000 kilometres, and can rise up again from there. During the uprise, the diamonds remain stable until they pass back through the transition zone from the lower to the upper mantle of the Earth, in which both the chemical equilibrium and the pressure and temperature conditions change again: here the minerals containing trivalent iron – perovskite and garnet – become unstable and release trivalent iron which then reacts with the diamonds. The carbon is re-oxidised in this process, the diamonds are destroyed and the trivalent iron is reduced to divalent iron. Carbonate melts can form again due to the presence of carbon dioxide, and the divalent iron is incorporated into the minerals olivine and pyroxene, which make up the majority of the upper mantle.
This enabled the researchers to show not only how and where carbonatite melts are formed in the Earth’s mantle. They also demonstrated how the carbon cycle from the Earth’s surface down to its interior functions: from carbon dioxide transported with carbonates into the interior, reduced to pure carbon and finally oxidised back to carbon dioxide as it rises. The resulting carbonatite melt, most of which is normally dissolved in a silicate melt at crustal pressures, ultimately brings the carbon dioxide back to the surface of the Earth through active volcanoes such as the Ol Doinyo Lengai.
More information: Rohrbach A &Schmidt M: Redox freezing and melting in the Earth’s deep mantle, Nature (2011), DOI:10.1038/nature09899, published online 23 March 2011.
Provided by ETH Zurich