Breakthrough in the search for the holy grail for data storage
(PhysOrg.com) -- One of The University of Nottingham’s leading young scientists has created a new compound which could lead to a breakthrough in the search for high performance computing techniques.
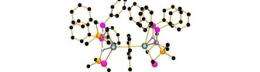
Dr Steve Liddle, an expert in molecular depleted uranium chemistry, has created a new molecule containing two Uranium atoms which, if kept at a very low temperature, will maintain its magnetism. This type of single-molecule magnet (SMM) has the potential to increase data storage capacity by many hundreds, even thousands of times — as a result huge volumes of data could be stored in tiny places.
Dr Liddle, a Royal Society University Research Fellow and Reader in the School of Chemistry, has received numerous accolades for his ground breaking research. His latest discovery has just been published in the journal Nature Chemistry.
Dr Liddle said: “This work is exciting because it suggests a new way of generating SMM behaviour and it shines a light on poorly understood uranium phenomena. It could help point the way to making scientific advances with more technologically amenable metals such as the lanthanides. The challenge now is to see if we can build bigger clusters to improve the blocking temperatures and apply this more generally.
Computer hard discs are made up of magnetic material which record digital signals. The smaller you can make these tiny magnets the more information you can store.
Although it may have somewhat negative PR it seems depleted Uranium — a by-product from uranium enrichment and of no use in nuclear applications because the radioactive component has been removed — could now hold some of the key to their research. Dr Liddle has shown that by linking more than one uranium atom together via a bridging toluene molecule SMM behaviour is exhibited.
He said: “At this stage it is too early to say where this research might lead but single-molecule magnets have been the subject of intense study because of their potential applications to make a step change in data storage capacity and realise high performance computing techniques such as quantum information processing and spintronics.”
Dr Liddle said: “The inherent properties of uranium place it between popularly researched transition and lanthanide metals and this means it has the best of both worlds. It is therefore an attractive candidate for SMM chemistry, but this has never been realised in polymetallic systems which is necessary to make them work at room temperature.”
Dr Liddle is a regular contributor to the School of Chemistry’s award winning Periodic Table of Videos — periodicvideos.com. The website, created by Brady Haran, the University’s film maker in residence, won the 2008 IChemE Petronas Award for excellence in education and training.
More information: A delocalized arene-bridged diuranium single-molecule magnet, Nature Chemistry (2011) doi:10.1038/nchem.1028
Abstract
Single-molecule magnets (SMMs) are compounds that, below a blocking temperature, exhibit stable magnetization purely of molecular origin, and not caused by long-range ordering of magnetic moments in the bulk. They thus show promise for applications such as data storage of ultra-high density. The stability of the magnetization increases with increasing ground-state spin and magnetic anisotropy. Transition-metal SMMs typically possess high-spin ground states, but insufficient magnetic anisotropies. Lanthanide SMMs exhibit large magnetic anisotropies, but building high-spin ground states is difficult because they tend to form ionic bonds that limit magnetic exchange coupling. In contrast, the significant covalent bonding and large spin–orbit contributions associated with uranium are particularly attractive for the development of improved SMMs. Here we report a delocalized arene-bridged diuranium SMM. This study demonstrates that arene-bridged polyuranium clusters can exhibit SMM behaviour without relying on the superexchange coupling of spins. This approach may lead to increased blocking temperatures.
Provided by University of Nottingham
















