Taking the guesswork out of understanding complex, multi-molecule transformations
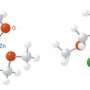
Multicomponent reactions (MCRs) that chemically combine three or more molecules into a brand new product are faster and generate less waste than traditional step-by-step synthetic procedures, making them invaluable in efforts to improve efficiency and sustainability. Since 1921, chemists have used an MCR called the Passerini reaction to produce bioactive, peptide-like chains made from three partners: carboxylic acids, carbonyl compounds, and cyanide-bearing molecules. However, a full understanding of this process has eluded researchers because its multipart workings are difficult to detect experimentally.
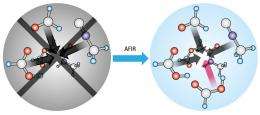
Now, Satoshi Maeda and Keiji Morokuma from Kyoto University and Shinsuke Komagawa and Masanobu Uchiyama from the RIKEN Advanced Science Institute in Wako have developed a computerized way to better identify the hidden mechanisms of one-pot, multi-step chemical transformations. Their technique, the artificial force induced reaction (AFIR), systematically squeezes and joins model compounds together in order to rapidly detect signatures of real MCR energy barriers.
According to Maeda, detailed theoretical understandings of MCRs have been scarce because most calculations require excellent estimates of transition state structures—intermediate and often highly strained geometric arrangements between molecules that correspond to the energetic peak of a reaction barrier. “Consequently, a trial-and-error process based only on [chemistry-based] intuition is unavoidable,” he says.
The AFIR method, on the other hand, requires no such presumptions. Maeda explains that when two molecules are pushed into each other with a weak force, they spontaneously relax into ‘dents’ in the potential barrier created by attractive electronic interactions between the reactants. By methodically pressing over all possible orientations, and inducing an artificial reaction from the relaxed positions, AFIR searches can identify every stable reaction pathway in a system.
To the team’s surprise, applying AFIR calculations to the Passerini reaction revealed that four components, not the long-thought three, must be involved (Fig. 1). Since the reaction barriers were so high, the researchers realized that an additional carboxylic acid—a known proton transfer catalyst—had to participate in the transition states leading to the final product. This finding should enable design of Passerini reactions with improved structural selectivity, notes Maeda.
Once perfected, the researchers anticipate their technique will make MCRs even more widespread. “Unlike standard methods, giving only pathways that are easy to find, the AFIR method has a unique ability to find unknown pathways in MCRs systematically,” says Maeda. “Information on reaction pathways is very important even for processes that do not normally occur, because chemists can initiate such reactions by controlling conditions, modifying substituents, and introducing new catalysts.”
More information: Maeda, S., et al. Finding reaction pathways for multicomponent reactions: The Passerini reaction is a four-component reaction. Angewandte Chemie International Edition 50, 644–649 (2011). onlinelibrary.wiley.com/doi/10 … e.201005336/abstract
Provided by RIKEN