Novel factor behind ES cells' neural default
Embryonic stem cells (ESCs) are highly regarded for their ability to give rise to the full range of cellular lineages found in the adult body, but left to their own devices ESCs tend to differentiate into neural lineages. Researchers from RIKEN have revealed how the nuclear protein Zfp521 is key to the default neural fate.
Embryonic stem cells (ESCs) are highly regarded for their ability to give rise to the full range of cellular lineages found in the adult body, a capacity known as pluripotency. In most cases, this differentiation needs to be steered by molecular cues that drive the stem cells’ progeny to adopt a specific fate, such as blood, muscle or bone. But left to their own devices, ESCs tend to differentiate into neural lineages. Indeed, in the absence of an inhibitory signal, such as BMP4, the preferred destination for ESC differentiation is neural ectoderm. This has been known for years, following demonstrations of the phenomenon in undifferentiated tissue in amphibians, but the mechanism behind this cell-intrinsic propensity has never been explained.
Daisuke Kamiya and colleagues in the Laboratory for Organogenesis and Neurogenesis, working in collaboration with the Laboratories for Stem cell Biology and Animal Resources and Genetic Engineering, have revealed how the nuclear protein Zfp521 is key to the default neural fate. In an article published in Nature, the group reports that this factor is both necessary and sufficient for driving the intrinsic neural differentiation of mouse ESCs.
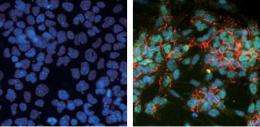
The study started from a GeneChip survey of mouse ES cells that had taken their first step down the road to neural differentiation, as revealed by GFP knocked into Sox1, a neural progenitor-specific marker. This turned up 104 genes of interest, 29 of which were even more interesting for their lack of expression in mesodermal tissues. Of these, Zfp521 was particularly attractive as it was found to be a potent promoter of neural differentiation when added to ESCs. Forced expression of Zfp521 led to increased expression of early neural markers such as Sox1, Sox3, and Ncad. The last of these, which encodes N-cadherin, is normally suppressed by the neural inhibitor BMP4, but Kamiya found that overexpression of Zfp521 overrode this effect. In situ hybridization showed that Zfp521 expression switches by around day 6.5 of development, and grows more pronounced in the neuroectodermal region of the epiblast, while it is absent in non-neural tissues.
To determine whether Zfp521 is truly necessary for neural differentiation of ESCs, Kamiya next knocked down its function by short hairpin RNA, resulting in a dramatic reduction of neural marker expressing cells. Importantly, this was not rescued by the addition of control cells, indicating that that action was cell autonomous. When the Zfp521-knockdown ESCs were injected into blastocysts, they failed to contribute significantly to the rostral neural tube, indicating a failure in primary neurulation.
Wanting to know the specific window of time in which Zfp521 exerts its function, the group examined its effects on a range of stage- and lineage-specific markers, from the earliest stages of undifferentiated ES cells, which reflects the developmental status of the inner cell mass (ICM), to various primary lineages differentiating from epiblast tissues. They found that while ICM development to epiblast was unaffected by loss of Zfp521 function, epiblast cells themselves failed to make the transition to neuroectoderm. Chromatin immunoprecipitation (ChIP) assays revealed that Zfp521 associates with a number of neuroectoderm-specific genes, but not with genes lined to the maintenance of pluripotency or differentiation into other lineages, suggesting that its effect is achieved by activating neural genes rather than inhibiting non-neural ones. This may be attributable to an interaction with a second factor, the histone acetylase p300, with which Zfp521 also associates. The model of default neural differentiation that emerges from these findings suggests that Zfp521 functions as a transactivator of neural specification, autonomously steering undifferentiated ESCs to adopt a neuroectodermal fate.
“This set of findings has given us insight into the mechanisms behind how a colony of undifferentiated cells such as ESCs can begin neural differentiation prompted early in development by Zfp521 in the absence of countering signals, such as BMP4,” says Sasai. “What we would like to work out next is whether this factor has similarly intrinsic effects in the epiblast as well.”
More information: Kamiya D, et al. 'Intrinsic transition of embryonic stem-cell differentiation into neural progenitors.' Nature. 2011 Feb 24;470(7335):503-9. Epub 2011 Feb 16. www.ncbi.nlm.nih.gov/entrez/qu … n&list_uids=21326203
Provided by RIKEN