Mechanical engineering at the molecular level: Self-assembly of nano-rotors (w/ Video)
German scientists from the Technische Universitaet Muenchen have managed to direct the self-assembly of rod-shaped molecules into rotors only few nanometers in size. The tiny systems serve the study of forces that act on molecules on surfaces and in cage-like structures. Their findings are published in the current online issue of the Proceedings of the National Academy of Sciences.
In the nanoworld many things are different. Scientists only recently started unveiling and harnessing the underlying laws and principles. A team associated with Professor Johannes Barth from the Physics Department of the TU Muenchen have now succeeded in capturing rod-shaped molecules in a two-dimensional network in such a way that they autonomously form small rotors that turn in their honeycomb-like cages.
Nature itself provides the role model for such self-organizing systems. This is how proteins bring reactants so close together that reactions can take place – reactions that are possible only in very close proximity. These effects are put to use in catalysts: surface reactants find their way to each other on the surface of these facilitators. However, the coveted dream of using self-organization effects in such a way that nano machines assemble themselves is still a thing of the future.
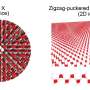
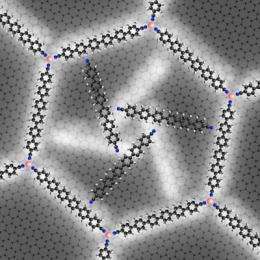
The rotors developed in Garching are an important step in this direction. First, the physicists built up an extensive nano lattice by allowing cobalt atoms and rod-shaped molecules of sexiphenyl-dicarbonitrile to react with each other on a silver surface. This results in a honeycomb-like lattice of extreme regularity with astonishing stability. Just like graphene, for which its discoverers were awarded the Nobel Prize only a few weeks ago, this lattice is exactly one atom thick.
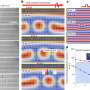
When the researchers added further molecular building blocks, the rods spontaneously gathered, typically in groups of three, in a honeycomb cell while neighboring cells remained empty. The chummy molecules must have had a reason for organizing themselves in threesomes. Under a scanning tunneling microscope the scientists were able to recognize why. The three molecules oriented themselves in such a way that the nitrogen ends each faced a phenyl-ring hydrogen atom. This triple-bladed rotor arrangement is so energetically advantageous that the molecules maintain this structure even when thermal energy drives it to rotation.
Because the honeycomb-cell is not round, but hexagonal, there are two different positions for the rotors that can be distinguished as a result of the interactions between the outer nitrogen atoms and the hydrogen atoms of the cell wall. Furthermore, the three molecules arrange in a clockwise and a counter-clockwise manner. In experiments at various carefully controlled temperatures the physicists were able to "freeze" all four states and examine them closely. They could thus determine the energy of these thresholds from the temperature at which the rotation resumed.
"We hope that in future we will be able to extend these simple mechanical models to optical or electronic switching," says Professor Johannes Barth. "We can set a specific cell size, we can specifically bring in further molecules and study their interaction with the surface and the cell wall. These self-organizing structures hold enormous potential."
More information: www.nature.com/nnano/journal/v … /nnano.2010.230.html
Provided by Technische Universitaet Muenchen