Finding genes that control mind and behavior
The highly sophisticated abilities of humans, such as memory, learning, cognition and thought, are achieved in the brain as a result of dramatic evolutionary development. Personality, preference, behavioral patterns, emotion and other mind-related functions also derive from brain functions. But what genes are responsible for the formation of the human brain and the remarkable abilities of the human mind? Jun Aruga, team leader of the Laboratory for Behavioral and Developmental Disorders at RIKEN’s Brain Science Institute (BSI), is working to identify the genes that play key roles in the brain, and to elucidate their functions by comparing genetic information in various organisms and conducting behavioral experiments in mice. His research is expected to clarify the origins of brain and nerve diseases and lead to the development of new therapies.
ZIC, a gene associated with brain anomalies
“When I was a high-school student, I was fascinated by the psychology books of Freud, Jung and others,” says Aruga. “Soon I became interested in the sophisticated abilities of the human brain, and so chose to study medicine at university.” After graduating from the School of Medicine, he joined the laboratory of Katsuhiko Mikoshiba at the Institute for Protein Research of Osaka University. Mikoshiba himself is now team leader of the Laboratory for Developmental Neurobiology at the BSI. “In those days, a technique for examining genes had just been established. I thought, what are the genes behind the outstanding potential of the human brain? Their functions are surely closely linked to the essential nature of humans. I wanted to identify the functions—that was my motivation for becoming a researcher,” says Aruga.
In Mikoshiba’s laboratory, researchers were studying the formation of the cerebellum, which performs important functions in motor learning and other aspects. “I was responsible for experiments for examining genes expressed in the process of cerebellar formation, and I discovered a gene that was expressed remarkably strongly. I became convinced that it must be important,” says Aruga. His intuition was spot-on. The gene, named ZIC by Aruga and others, proved to play a critical role in the formation of the entire brain, not just the cerebellum.
The formation of the brain begins with the generation of tabular neural tissue known as the neural plate, which undergoes rounding to form a tubular structure called the neural tube. “The rounded neural plate closes in its center, then other portions close like a zipper moving back and forth, resulting in completion of the neural tube closure. One end of the neural tube grows gradually into the complex brain.”
Aruga generated mice that lack the ZIC gene and found that they had malformed neural tubes with incomplete closure of the anterior (forward) or posterior (rearward) end. He also found other malformations, including union of the left and right cerebral ventricles, known as holoprosencephaly, and various cerebellar malformations.
A group of researchers in the USA also subsequently demonstrated through human gene analysis that mutations of the human ZIC gene are associated with holoprosencephaly and cerebellar malformations. “Mutations of the same ZIC gene were found to cause similar brain malformations in humans and mice.” More recently, mutations of the ZIC gene have been shown to cause not only brain malformations, but also anomalies in the left-right positions of internal organs.
SLITRK1, a gene suppressing anxiety/depression susceptibility
The ZIC gene also serves to control the expression of other genes. “Humans and mice have five kinds of ZIC genes. In 2003, we discovered that some of them control the expression of SLITRK.” SLITRK is a gene for a membrane protein that penetrates biomembranes and cell membranes wrapping cell components (organelles). “In the brain, neurons extend neurites—fine filaments called axons or dendrites—to each other to exchange information. The membrane protein produced by SLITRK is structurally similar to the protein involved in the extension of neurites, so I thought this gene must also be important, and continued the research.”
In 2005, a research group in the USA announced that SLITRK1, one of the six SLITRK genes in humans and mice, is associated with Tourette’s syndrome — a disorder, commonly developed in childhood, that causes persistent motor and vocal ‘tics’. It was one of the first discoveries of causal genes for the disease. “We succeeded in preparing mice that lacked the SLITRK1 gene,” says Aruga. “Surprisingly, the mice looked normal at first glance. Mice lacking ZIC have obvious malformations, and many of them die soon after birth. Mice lacking SLITRK1, however, can grow to maturity and females can even bear offspring.”
Aruga and others then decided to conduct behavioral experiments to examine the properties of SLITRK1-deficient mice in detail. “Behavioral experiments are very effective for examining brain functions. We asked the BSI’s Research Resources Center to perform behavioral experiments—they conduct experiments for any laboratory at RIKEN on request, which is very helpful for some of the smaller laboratories because some of the experiments can take up to two months. Kei-ichi Katayama, a fellow researcher in my laboratory, also conducted extensive additional experiments to construct a comprehensive profile of the behavioral abnormalities in mice lacking SLITRK1.”
The behavioral experiments yielded two interesting findings. One was obtained through an experiment in which a mouse is placed in the center of an elevated cross-shaped platform having walls on only one of the two crossed arms (Fig. 1). A normal mouse will generally move around the platform to explore its situation. The SLITRK1-deficient mice, however, tended to explore the wall-less limbs less frequently. “Many studies have shown that mice with a tendency for anxiety use the wall-less limb less frequently in this test.”
The other finding came from an experiment in which a mouse is suspended by its tail. Normal mice struggle desperately at first, and soon become still, periodically taking up the struggle again. “The SLITRK1-deficient mice were found to rest in the quiescent state for much longer. They are also known to have a more of a tendency for depression.”
Various other behavioral experiments have revealed that the SLITRK1-deficient mice are highly susceptible to anxiety and depression. Tourette’s syndrome in humans has also been suggested to be associated with a tendency for anxiety and depression.
‘Playing catch’ with clinical researchers
Research then turned to what is happening in the brain of SLITRK1-deficient mice. Niall Murphy, who at the time was serving as a unit leader at the BSI (and who is currently at the University of California), took on the challenge of exploring the neurological effects of SLITRK1 deficiency. “Dr Murphy found that noradrenaline increased in the frontal lobe of the brain in such mice,” says Aruga. Noradrenaline is a neurotransmitter responsible for signaling among neurons and is produced in neurons in a particular region of the brain. These neurons extend neurites throughout the brain and secrete neurotransmitters to regulate the function of the brain as a whole, influencing moods such as comfort and discomfort, and behavioral patterns.
“In behavioral experiments using SLITRK1-deficient mice, clonidine—a drug that regulates noradrenaline—was found to alleviate anxiety. Clonidine is now used as a therapeutic agent for Tourette’s syndrome in humans.” These findings suggest that SLITRK1-deficient mice are useful as an animal model for Tourette’s syndrome and are likely help in elucidating the origins of the disorder, which could lead to the development of new therapies.
“We are right now engaged in an exploratory study using mice and other laboratory animals in a search for genes that are intimately involved in brain formation and functions, like ZIC and SLITRK1. We would then pass any discoveries to clinical researchers, who could examine the relationship between the gene and disease in humans. We would in turn examine in more detail the functions of the gene in laboratory animals on the basis of the clinical information. By ‘playing catch’ with clinical researchers in this way, our research will help to clarify the causes of human diseases and develop therapies.”
Aruga and others have already begun ‘playing catch’ with other groups. “In 2009, a clinical researcher presented a paper reporting on an association between the SLITRK2 gene and manic–depressive psychosis. We generated mice deficient in the gene and examined their behavior. The mice were found to be highly active, and exhibited a tendency similar to mania.”
Joint research with Murphy has shown that SLITRK2-deficient mice have altered serotonin concentrations in the hippocampus and amygdaloid body, areas of the brain related to memory and emotion, respectively. “Since this change in serotonin content is considered to be associated with active behavior, we tried administering lithium and other drugs used to prevent the manic state in human manic–depressive psychosis. But the mania-like tendency was not alleviated. We are now conducting a demonstration study to determine whether the SLITRK2 gene and manic–depressive psychosis are in fact correlated at all.”
SLITRK6, a gene involved in the formation of the neural circuit of the inner ear

More recently, in November 2009, a group of researchers including Aruga, Katayama and Azel Zine from the University of Montpellier in France discovered that the SLITRK6 gene plays a key role in the formation of the sound-sensing neural circuit of the inner ear. “Mice deficient in SLITRK6, generated by Katayama, looked normal in a similar way to the SLITRK1-deficient mice. At that time, Prof. Zine was working in my laboratory as a visiting researcher. Although with us for only two weeks, he astonished us just before returning home by announcing that he had made a discovery in mice lacking the SLITRK6 gene.”
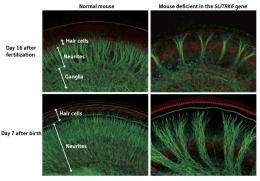
Zine is a researcher who specializes in the inner ear. The cochlea at the heart of the inner ear contains hair cells that sense sound. Neurons extend neurites to the cochlea to transmit the sound information captured by the hair cells to the brain (Fig. 2). “Prof. Zine discovered that the number of neurites in SLITRK6-deficient mice is lower than in normal mice.”
Aruga’s research then allowed the puzzle to be pieced together. During neural circuit formation, the cochlear hair cells produce neurotrophin, a protein that promotes neuron development. This secretion stimulates the growth of neurites. Aruga demonstrated that in SLITRK6-deficient mice, the secretion of neurotrophin is suppressed. “Lower neurotrophin seems to reduce the number of neurites formed. In fact, when we manually applied neurotrophin to ganglial neurons, the number of neurites returned to close to normal levels.” The findings are expected to help in the development of therapies for inner ear dysfunction.
Genome exploration

“The ZIC and SLITRK genes were discovered by intuition and the accumulation of knowledge. We have begun a research project to search for important genes like these by comprehensively and systematically exploring genetic information,” says Aruga. “In the twenty-first century, many projects aimed at decoding the genomes of a wide variety of organisms have been established and many have been completed. When I set up my laboratory at RIKEN in 2004, I wanted to do research using a revolutionary approach that has become possible only recently. I thought it might be possible to identify the genes that are important to the human brain by comparing the genomes of a range of organisms.” Genetic information is written in the arrangement of the four bases (thymine, adenine, guanine and cytosine) in DNA, and the genes of different organisms can be compared on the basis of the similarity of this arrangement. For the ZIC genes, for example (Fig. 3), monkeys are similar to humans in almost all regions. Mice and rats are also similarity in many regions, while chickens, toads and blowfish are similar to humans in only a few regions. The base sequences of these shared regions can therefore be considered to be highly conserved in the process of evolution, and are therefore likely to carry information or functionality that is important for vertebrates.
In his exploration of genomes, Aruga took particular note of the leucine-rich repeat (LRR), part of the structure of membrane proteins (Fig. 4). “Membrane proteins with the LRR structure vary greatly among multicellular animals having nervous systems, such as drosophila and nematodes, with a particularly wide variety in vertebrates. Many LRR-membrane proteins are primarily expressed in brain cells. We conjecture that the LRR structure has played a critical role in the process of evolution that has lead to increased brain size, as in humans.”
In both humans and mice, there are almost 100 membrane proteins with the LRR structure, include some suggested to be associated with psychiatric diseases such as schizophrenia, temporal lobe epilepsy and obsessive–compulsive neurosis. The membrane proteins produced by the SLITRK genes also have the LRR structure. “We are investigating membrane proteins with the LRR structure but with unknown function.”
Aruga’s group is conducting this study while ‘playing catch’ with the BSI’s Laboratory for Molecular Psychiatry, headed by Takeo Yoshikawa, which is pursuing research on the association of human genes and psychiatric diseases. “Prof. Yoshikawa and others are working to determine whether our target genes are related to human psychiatric diseases. Conversely, we are conducting extensive studies to determine whether a genetic mutation found in a patient with psychiatric disease by Yoshikawa is linked to the condition, through behavioral experiments on mice deficient in the gene we have created. We are ‘playing catch’ within the BSI to contribute to medicine. Ultimately, I want to identify a gene that is closely related to the essential nature of being human. I want to find a convincing answer to the question that has fascinated me since I was a high-school student.”