Paper highlight: Solvent-mediated end-to-end assembly of gold nanorods
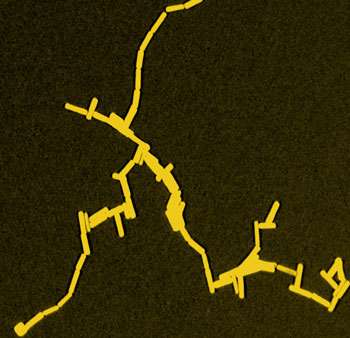
Argonne National Laboratory scientists have developed a new method for the controlled end-to-end assembly of rod-shaped gold nanoparticles.
The nanoparticles are synthesized chemically using an established technique, producing high-quality gold nanorods with lengths of approximately 70 nm and diameters of approximately 15 nm, stabilized in water by hydrophilic surfactant molecules.
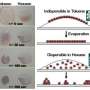
The sides of the rods are better protected by the surfactants than the ends, so that when additional ligand molecules that bind to gold are added to the solution, they preferentially attach to the ends of the rods.
If these ligands are hydrophobic, then the rods will spontaneously organize end to end when they are placed in an appropriate mixture of organic solvents and water.
Analysis of assembly kinetics showed that this solvent-mediated process is very different from the assembly processes that have previously been used to arrange nanorods, such as covalent bonding and hydrogen bonding.
The analysis also demonstrates the conditions necessary for obtaining selective end-to-end assembly; in particular, it was found that nanorod samples need to be aged for several days between synthesis and assembly.
The results thus provide a new means of arranging anisotropic nanoparticles into controlled configurations, allowing for the bottom-up production of functional nanomaterials.
More information: Y. Wang, A.E. DePrince III, S.K. Gray, X.-M. Lin, and M. Pelton, J. Phys. Chem. Lett., 1, 2692 (2010)
Provided by Argonne National Laboratory