Scientists invent first technique for producing promising anti-leukemia agent
Kapakahines, marine-derived natural products isolated from a South Pacific sponge in trace quantities, have shown anti-leukemia potential, but studies have been all but stalled by kapakahines' lack of availability.
But using only acetylene gas, a handful of amino acids, and a dozen inventive steps, a team from The Scripps Research Institute has finally established the first technique to synthesize kapakahines in the laboratory in large quantities, more than a decade after their discovery. With supplies now in hand, and unlimited production potential established, research on the compound can proceed and may eventually lead to new drug treatments.
The research is described in a paper published online by the Journal of the American Chemical Society on April 17, 2009.
Cripbrochalina olemda appears to the uninitiated as a common tube-type sponge similar to countless others you might find on reefs throughout the tropics. But this species, discovered in 1995, is one of a growing number of marine organisms researchers have found that naturally produce chemicals with great potential for fighting diseases such as cancer.
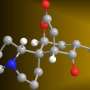
C. olemda produces a compound called kapakahine B, among other molecules of interest, that has shown potential for fighting leukemia. As important, kapakahine B, named after the Hawaiian word kapakahi, which means "twisted," has an unusual structure that researchers had never seen before, suggesting that its ability to fight cancer cells may stem from some never-before-seen mechanism.
The problem is that amassing enough of the kapakahines to conduct full studies had proven an untenable proposition. Each sponge contains only a relative speck of the compounds. Even if massive quantities of the sponge could be harvested—devastating ocean ecosystems in the process—it would still be difficult to get enough material to work with, and would likely be impossible to get enough for commercial use should a kapakahine prove an effective disease treatment. Being able to synthesize the compounds in the lab would solve the problem, but this has proven challenging.
"Chemists are always attracted to things that are bizarre," says Phil Baran, a Scripps Research chemist and leader of the group, of the kapakahines' strange twists. While at times that can be enough motivation for research, in this case, the attraction is deeper. "There is no shortage of biologists who want to look at active molecules, but if you can't provide the molecule, then they can't go very far."
Groups of chemists around the world have endeavored unsuccessfully to devise a method for synthesizing the kapakahines. The Scripps Research team's success with the challenge began with more basic research to synthesize a simpler related compound with no known pharmaceutical potential. Tim Newhouse, a graduate student in the Scripps Research Kellogg School of Science and Technology, last year published with Baran a paper detailing his invention of a simple and highly efficient synthesis of a complex alkaloid called psychotrimine, which was originally isolated from a rainforest shrub.
Risky Chemistry
The Newhouse synthesis hinged on the creation of a highly reactive and selective chemical component referred to as a quaternary center that, because of structural similarities, also drives the essential first step in the kapakahines synthesis. Chad Lewis, a postdoctoral researcher in the Baran lab, then teamed up with Newhouse to set out on a somewhat riskier venture to develop a second stage needed to synthesize kapakahines.
On paper, by analyzing and deconstructing the kapakines' structure, the Baran group predicted that using the quaternary center they could produce two intermediate isomers, or molecules with the same chemical formula but different structures. One isomer, they predicted, would be easy to make, but would be a divergence from the chemical pathway to the kapakahines. The other would be an ideal stepping stone toward the kapakahines, but more difficult to make, meaning in this case only the smallest of quantities would be produced relative to the first isomer. But the second isomer would be much more reactive, and in theory its concentration would grow sufficiently as it moved toward equilibrium with the first isomer.
The risk was that substantial work was required to discover whether the isomers would behave as predicted, allowing the synthesis to proceed. If they didn't, all the work would have led to a dead end. "It was a bit of a dare because this was just a paper idea," says Baran, "It was the kind of thing that we knew would be shocking if it actually worked."
The researchers' predictions did ultimately prove correct, allowing them to synthesize two kapakahines for the first time and in gram quantities. That means that now, some 14 years after they were discovered, full research into the kapakahines' potential can finally proceed.
One of the compounds, kapakahine B, has shown potential in fighting leukemia cells. Interestingly, the second, kapakahine F, which has a very similar structure, shows no such activity. The only difference between the two is the addition on B of a single amino acid residue.
Having this critical component already identified should simplify studies of kapakahine B's anti-leukemia activity, an essential step in research on any potential drug treatment. And, because the kapakahine structure is novel, there is a good chance that this activity is different from that seen in other compounds with potential against leukemia, opening the possibility of an entirely new form of treatment.
Another tantalizing prospect is that the relatively inactive kapakahine F could be easily manipulated to form a library of new compounds by adding different chemical groups to the reactive spot where phenylalanine leads to kapakahine B's activity, and these analogs could proven even better at fighting leukemia or other forms of cancer than B.
More information: The study, authored by Newhouse, Lewis, and Baran of Scripps Research, is titled "Enantiospecific Total Syntheses of Kapakahines B and F." For more information, see pubs.acs.org/doi/abs/10.1021/ja901573x
Source: The Scripps Research Institute (news : web)