How a Cell's Mitotic Motors Direct Key Life Processes
University of Massachusetts Amherst biologists have discovered a secret of how cells organize chromosomes to prepare for dividing. Their unexpected finding is reported in this week’s issue of the journal, Current Biology.
The experiments sought to reveal how the cell’s tiny, two-part chemical engine known as dynein, just 40 nanometers in diameter, takes charge of mitosis and keeps the delicate strands of chromosomes in order and in position. Until now, cell biologists had assumed it was the dynein’s cargo domain that regulated this process. UMass Amherst cell biologist Wei-lih Lee and colleagues showed that it is the motor domain instead.
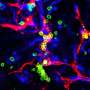
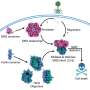
Dynein, like a delivery truck, carries cargo, Lee explains, but this protein truck is specialized because it interacts chemically and physically with the road. In the cell, this means dynein travels along segments of polymeric microtubule “roads” that grow and shrink as needed by adding or dropping sections. From experiments in budding yeast, Lee, with a talented postdoctoral fellow, Steven Markus, and biology junior fellow Jesse Punch, found that “dynein has a preference for locating at the ends of these microtubule tracks.”
Lee says a lot of credit for a cleverly designed and executed set of experiments goes to Markus, who cut the dynein engines into motor and cargo halves and challenged the yeast cells to divide with access to only one part of the protein at a time. Markus also designed brighter-than-usual fluorescent probes to attach to the two dynein parts, red for the engine, green for the cargo domain. The strategies worked. The UMass Amherst research team now has one of the most elegant and practical probes for studying dynein function. Lee says, “I’m already getting requests from other researchers who want to use our new probes.”
In this system, they observed that like a moving walkway at the airport, “dynein is a smart truck because it parks at the end of the microtubule, and ‘rides’ along as the track grows,” Lee explains. “Our findings show that the dynein’s motor domain, the engine’s core, is responsible for this end-binding property, which is surprising given that the same domain is used for walking along the track.”
Applying their new understanding to cell division, the researchers say, “our findings further suggest that the dynein engine is turned off when it is parked on the microtubule end, but then turned on upon reaching the proper attachment site in the daughter cell’s wall,” says Lee. “This mechanism allows the yeast cell to control dynein activation with high accuracy” and avoids potential problems of transporting an “activated” protein through the cell.
Results of this new knowledge in basic science are also relevant for human nerve cell function. “It has already been shown that nerve cells use the same mechanism as yeast does to move the cell body,” says Lee. Dynein malfunction can lead to mistakes in nerve cell migration which causes poor brain development disease such as lissencephaly.
Provided by University of Massachusetts Amherst