First functional stem-cell niche model created
Like it or not, your living room probably says a lot about you. Given a few uninterrupted moments to poke around, a stranger could probably get a pretty good idea of your likes and dislikes, and maybe even your future plans. Scientists at the Stanford University School of Medicine employing a similar "peeping Tom" tactic to learn more about how stem cells develop have taken a significant step forward by devising a way to recreate the cells' lair — a microenvironment called a niche — in an adult animal.
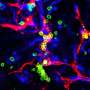
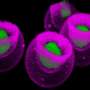
"We have isolated the cells in mouse bone that make bone and cartilage from scratch and attract wandering blood stem cells," said Irving Weissman, MD, the Virginia & D.K. Ludwig Professor for Clinical Investigation in Cancer Research and the director of Stanford's Institute for Stem Cell Biology and Regenerative Medicine. "The stem cells can and do settle in these 'niches' and make blood that is exported to the body."
The research marks the first time that scientists have successfully recreated a functional stem-cell niche for further study. Weissman and his colleagues plan to use the model system to determine how the niche environment interacts with the blood stem cells to affect their development and fate, and how leukemias respond to these niches. They will also investigate the bone and cartilage healing capacity of these cells.
Weissman is the senior author of the study, which will be published Dec.10 in the advance online issue of Nature. Graduate student Charles Chan shares first authorship with postdoctoral scholars Ching-Cheng Chen, PhD, and Cynthia Luppen, PhD.
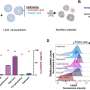
Blood-forming stem cells typically reside in the bone marrow. The researchers found that a specific subset of fetal mouse bone cells could not only take up residence and produce bone when injected near the kidney of an adult animal, but they also generated a bone marrow cavity that sheltered host-derived blood stem cells. In contrast, other subsets of fetal bone cells generated only bone.
"An amazing part of this study was the formation of organized bone, cartilage and blood stem cell niches from an initially dispersed set of cells," said Weissman, who is also a member of the Stanford Cancer Center. "If we can find the daughter cell in this population that is responsible for niche formation, we may learn enough to eventually be able to expand blood stem cell numbers so that a small number, say from umbilical cord blood, can be made into enough to treat several patients with failure of blood formation."
Suppressing the expression of factors involved in a specialized bone-building process called endochondrial ossification in the host mouse stopped the formation of the marrow cavity and the recruitment of host stem cells. Using similar fetal bone cells from parts of the skeleton that do not undergo the process — such as the skull and the jaw — also blocks cavity formation. The findings suggest that endochondrial ossification is a necessary step in setting up house for stem cells.
Source: Stanford University