Seeing Nanotubes Targeting Tumors In Vivo
Carbon nanotubes have significant potential for delivering both imaging and therapeutic agents to tumors, but there is still a need to better quantify how well these rolled-up sheets of graphite can target tumors. Now, thanks to the development of a microscope capable of measuring Raman spectroscopic signals from living mice, researchers have a noninvasive tool to study where carbon nanotubes travel once they are injected into the blood stream.
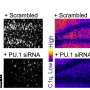
Reporting its work in the journal Nano Letters, a team of investigators led by Sanjiv Gambhir, M.D., Ph.D., principal investigator of the Center for Cancer Nanotechnology Excellence Focused on Therapy Response (CCNE-TR), based at Stanford University, and Hongjie Dai, Ph.D., also a member of the CCNE-TR, described its use of an optimized Raman microscope to track two different sets of carbon nanotubes as they transited through the body of living mice. One of the nanotubes was covered with the tumor-targeting peptide known as RGD; the other set was used without any added functionality.
Although other investigators have used positron emission tomography (PET) to follow radioactively labeled nanotubes as they move through the body, this technique requires the use of expensive radioisotopes and scanning instruments. To overcome these limitations, the CCNE-TR team took advantage of the fact that carbon nanotubes generate a characteristic Raman emission peak. Earlier this year (click here to see story), Dr. Gambhir and his colleagues described a new type of Raman microscope designed specifically for use in bioimaging studies.
Using this Raman microscope, the investigators were able to track differences in nanotube trafficking between the targeted and untargeted nanotubes. Although both sets of nanotubes showed an initial spike in tumor accumulation, the concentration of untargeted nanotubes in tumors began dropping as early as 20 minutes after injection. In contrast, the tumor concentration of the targeted nanotubes remained elevated for at least 72 hours after injection. In animals treated with the targeted nanotubes, tumors were readily visible as early as 2 hours postinjection and for at least 72 hours. The investigators noted that their results are consistent with those obtained using radioactively labeled nanotubes and PET imaging.
This work, which is detailed in the paper “Noninvasive Raman Spectroscopy in Living Mice for Evaluation of Tumor Targeting With Carbon Nanotubes,” was supported by the NCI Alliance for Nanotechnology in Cancer, a comprehensive initiative designed to accelerate the application of nanotechnology to the prevention, diagnosis, and treatment of cancer. An abstract of this paper is available at the journal’s Web site.
Provided by National Cancer Institute