New way of inhibiting cell cycle shows promise
Geneva, Switzerland: A new anti-cancer compound that works by blocking a part of the cell's machinery that is crucial for cell division has shown promising results in a phase I clinical trial in patients who have failed to respond to other treatments. Now it is going forward into a phase II clinical trial programme. In addition, the compound will also be tested in combination with other anti-cancer drugs to see whether combined therapies could be even more effective.
Professor Patrick Schöffski told the 20th EORTC-NCI-AACR Symposium on Molecular Targets and Cancer Therapeutics in Geneva today (Wednesday 22 October) that after 50 patients had been given the compound BI 6727 in doses ranging from 12 to 450 mg, two patients with advanced bladder and ovarian cancers had shown confirmed partial responses and a further 32% of the patients had stable disease.
"The results so far indicate that BI 6727 is well tolerated by patients, with no serious side-effects detected. We have observed encouraging anti-tumour activity, which we would not necessarily expect to see in a phase I trial, and which warrants investigation in further clinical trials," said Prof Schöffski, who is professor of medical oncology and head of the Department of General Medical Oncology at the University Hospitals Leuven (Belgium).
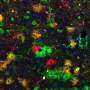
BI 6727 is one in a series of compounds developed by Boehringer Ingelheim that work by inhibiting the action of a protein called Polo-like kinase 1 (Plk1), high levels of which are present in human tumours, but not in normal tissue. Plk1 is involved in cell growth; inhibiting it leads to abnormal mitotic spindles – the structures that separate the chromosomes into daughter cells during cell division – and this disrupts cell division, inhibiting the growth of tumour tissue.
"Plk1 inhibitors are targeted, cell cycle blockers that lead to spindle defects by inhibiting a key regulator of mitosis. They act on the mitotic spindle in a completely different manner compared to established anti-cancer agents such as vinca alkaloids or taxanes that directly bind to structural components of the mitotic spindle. Due to high levels of Plk1 in tumour cells compared to surrounding healthy tissue, compounds such as BI 6727 are effectively targeting dividing cancer cells," said Prof Schöffski. "The results from this phase I trial suggest that BI 6727 potentially is a 'first in class' Plk1 inhibitor and the anti-tumour activity we have seen supports Plk1 as a therapeutic target."
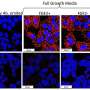
Preclinical data also presented at the meeting (abstract no: 430) show highly selective target inhibition and cellular activity at very low concentrations for this compound. BI 6727 shows excellent efficacy in multiple xenograft models of human cancer and its distinguishing features are its pharmacokinetic characteristics (what the body does to the drug) that allow for long-lasting tumour exposure.
The objectives of the phase I trial had been to assess the maximum tolerated dose of BI 6727 and its overall safety, the pharmacokinetics, and preliminary efficacy. Each treatment consisted of a single, one-hour infusion of the drug and it was given to sequential groups of three to six patients with advanced or metastatic solid tumours. If the cancer did not progress, further treatments were given at three-week intervals. The average number of treatments per patient was four, and at least 16 courses of treatment were given to two patients. The maximum tolerated dose was 400 mg.
The main adverse side effects of the drug were blood-related. Reduced white blood cells (neutropenia) or platelets (thrombocytopenia) were a result of Plk1 inhibition in normal cells and were both treatable and reversible events. About ten per cent of patients suffered from fatigue, mostly mild to moderate.
Speaking in September before the symposium started, Prof Schöffski said: "I will present the most up-to-date responses at the Geneva meeting, but, so far, one patient with urothelial [bladder] cancer has had clinical benefit for more than 16 cycles with BI 6727 and achieved confirmed partial response with the tumour shrinking by 42% within four cycles. Previously, this patient had failed other standard and experimental treatments. The cancer has not progressed since BI 6727 therapy started.
"A second patient with ovarian cancer had a confirmed partial response after two and four cycles, but her disease progressed at the sixth cycle. Previously, she had been treated with several courses of standard ovarian cancer treatments, including taxanes and cisplatinum.
"The initial part of the trial is completed, and now we have recruited a further 12 patients to compare different durations of infusion of the drug. We expect to have results from this soon."
Prof Schöffski, who will be giving an invited "state of the science" lecture on Plk1 at the Geneva symposium, said the results for BI 6727 so far were promising. "This agent is among the few Polo-like kinase inhibitors in early clinical development. Boehringer Ingelheim has advanced this compound from its Plk1 inhibitor portfolio into phase II, due to favourable pharmacology and the promising safety and efficacy seen in this phase I trial."
Source: ECCO-the European CanCer Organisation