Study on salmonella self-destruction
ETH Zurich biologists, led by Professors Martin Ackermann and Wolf-Dietrich Hardt, in collaboration with Michael Doebeli of the University of British Colombia in Vancouver (CN), have been able to describe how random molecular processes during cell division allow some cells to engage in a self-destructive act to generate a greater common good, thereby improving the situation of the surviving siblings.
The biologists investigated this unusual biological concept using the pathogenic salmo-nella bacteria as an example. Diseases caused by salmonellae are very unpleasant and even life-threatening. When contaminated food is consumed – for example, egg-based foods or chicken and meat – salmonella bacteria enter the gastro-intestinal tract where it triggers infection. Vomiting and diarrhoea can last for days.
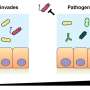
Normally, salmonellae grow poorly in the intestine because they are not competitive with other bacteria of the gut. However, this dynamic changes if salmonellae induce an in-flammatory response, namely diarrhoea, which suppresses the other bacteria. The in-flammation is triggered by salmonellae penetrating into the intestinal tissues. Once in-side, salmonellae is killed by the immune system. This in turn creates a conflict: salmo-nellae are either suppressed by the other bacteria in the gut, or die while trying to elimi-nate these competitors.
As Ackermann, Hardt and Doebeli report, salmonellae have found a surprising solution to this conflict. Inside the gut, the samonella bacteria forms two groups that engage in job-sharing. A first group invades the tissue, triggers an inflammation, then dies. A sec-ond group waits inside the gut until the inactivation of the normal intestinal flora gives them an opportunity to strike.This second group then multiplies unhindered.
Random processes and self-sacrifice
What determines whether an individual salmonella bacterium cell self-sacrifices, or whether it will wait and benefit from the sacrifice of others? The two groups are clones of the same genotype, so genetic differences do not play a role. Rather, the difference between the two groups is a result of random molecular processes during cell division. Cellular components are randomly distributed between the two daughter cells with each cell receiving a different amount. The resulting imbalance can be amplified and lead to different properties of the clonal siblings.
In recent years it has been recognized that such random processes in a cell can have a large influence on individual cells. The work by the ETH Zurich researchers reveals a new biological explanation for this phenomenon. The two salmonella phenotypes share their work, with the result being that they achieve what a single phenotype on its own would not be capable of doing. This scenario is fundamentally different from the usual explanations and presupposes that individual phenotypes interact and have an effect on one another. The self-sacrifice of phenotypes may be quite common among pathogenic bacteria, for example, among the pathogens causing diarrhoea after antibiotic treatment (clostridia) or pneumonia (streptococci).
Essential findings
Professor Ackermann says that "Random processes could promote job-sharing in many different types of organisms." Many bacteria manufacture substances which are toxic to their hosts but which are only released into the host environment if the bacteria sacrifice themselves - if this is the sole method to get the toxin out of the cell. This is why every cell makes a decision: toxin and death or no toxin.
He stresses that it would not have been possible to study this theory so thoroughly with-out the collaboration that took place among the three specialist groups: Professor Hardt's group specialises in salmonella infections; Professor Doebeli is a mathematician and theoretical biologist; and Professor Ackermann's group focuses on phenotypic noise.
Source: ETH Zurich/Swiss Federal Institute of Technology