Accumulated bits of a cell's own DNA can trigger autoimmune disease
A security system wired within every cell to detect the presence of rogue viral DNA can sometimes go awry, triggering an autoimmune response to single-stranded bits of the cell's own DNA, according to a report in the August 22nd issue of the journal Cell, a Cell Press publication. The source of that single-stranded DNA is so-called endogenous retroelements—genetic elements accounting for a substantial portion of the genome that can move to new locations using a "copy and paste" mechanism, according to the researchers.
The new findings help to explain the cause of a rare autoimmune disorder known as Aicardi-Goutieres Syndrome in which infants appear to suffer from an acute viral infection, despite the fact that no virus had ever been found.
" We and others had demonstrated the existence of a DNA detection pathway within cells, but we are still early in our understanding," said Daniel Stetson of the Howard Hughes Medical Institute and the University of Washington, Seattle. "Our findings offer an important piece of evidence that this pathway is not only very relevant, but it can be the cause of severe autoimmune disease."
Detection of foreign nucleic acids is an ancient form of host defense, the researchers explained. In vertebrates, nucleic acid detection activates a program of antiviral defense designed to neutralize the spread of infection. This antiviral program is coordinated by type I interferons (IFNs), which direct a multifaceted response to restrict viral replication within infected cells, alert neighboring cells to the presence of infection, and expand white blood cells to provide long-term and specific protection against the virus.
The defense mechanism includes two systems: one consisting of "Toll-like receptors" on specialized, sentinel immune cells that monitor for infection and another that detects viral nucleic acids within the infected cell itself. That internal system includes one arm for detecting RNA and another for detecting DNA.
The biological relevance of those internal DNA sensors remained somewhat mysterious, according to Stetson, because the "nuts and bolts" of the system hadn't been worked out.
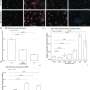
In a screen for proteins relevant to this pathway they call the interferon-stimulatory DNA (ISD) response, the researchers now identify an enzyme known as 3' repair exonuclease, aka Trex1. In studies of mice, the researchers showed that single-stranded DNA fragments derived from endogenous retroelements accumulate in Trex1-deficient hearts. Those fragments are produced through a process known as reverse transcription in which specialized enzymes copy RNA back into single-stranded DNA. Trex1 usually breaks down reverse-transcribed DNA of those endogenous retroelements, keeping the ISD response in check.
Mutations in the human Trex1 gene were already known to cause Aicardi-Goutieres Syndrome, although the mechanism remained uncertain. The new findings suggest that the syndrome is triggered by an accumulation of reverse-transcribed DNA. "In a sense, it's an enemy from within," Stetson said.
A similar mechanism may underlie other immune disorders as well, he added. In fact, other mutations in Trex1 have been linked to an autoimmune disorder called chilblain lupus and are found more frequently in people with systemic lupus erythematosus than in healthy individuals.
The findings also suggest an unanticipated contribution of endogenous retroelements to autoimmunity.
" Just as commensal bacteria outnumber our own cells by four or five orders of magnitude, endogenous retroelements outnumber our genes by at least 100-fold," the researchers concluded. "Both have the potential to be detected by the immune system and cause autoimmune disease. Therefore, specific mechanisms evolved to prevent this, and Trex1 represents the first example of a mechanism to prevent autoimmunity caused by endogenous retroelements."
Source: Cell Press