New molecular imaging techniques may lead to advances in disease treatment
A promising new technique has been developed that will enable more accurate non-invasive positron emission tomography (PET) imaging of new cells injected into the body, according to researchers at SNM's 55th Annual Meeting. The new technique, which involves engineering antibody fragments to act as reporter genes—or markers that signal cells of interest for PET imaging purposes—could significantly advance the study of genetically engineered cells to treat diseases.
"Genetic cell engineering is the focus of intense research in almost all areas of medicine and shows great promise for treatment of common illnesses such as heart disease, diabetes, and Parkinson's disease and other neurodegenerative disorders," said Wolfgang Weber, lead researcher of the study, Cell Surface Expression of an Engineered Antibody as a PET Reporter Gene for In Vivo PET Imaging, which was performed at the Department of Molecular and Medical Pharmacology at UCLA in collaboration with the Department of Chemistry at UC Davis.
However, despite intense efforts, researchers have few solid, noninvasive methods for accurately tracking the location, function and viability of small numbers of transplanted cells. "Our research shows that using antibodies as reporter genes in PET imaging provides these capabilities and could contribute to improved treatment of a number of potentially devastating diseases," added Weber, now professor of nuclear medicine at the University of Freiburg, Germany.
To improve PET imaging in this area, researchers have been studying the use of reporter gene–probe combinations. With this technique, cells are created to synthesize a protein that binds to or metabolizes radioactive reporter probes that are injected into the body and detected with PET imaging technology. However, most available reporter gene combinations are not aptly sensitive or specific and have significant limitations in terms of tracking the cells of interest to researchers.
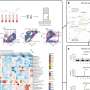
In this new research, Weber and his team explored using cell surface–bound antibody fragments as reporter genes. These engineered antibody fragments, developed by the group of Claude Meares at Davis, bind irreversibly to low-molecular-weight antigens, which act as reporter probes. Cell culture and animal studies demonstrated intense and highly specific uptake of the probes in cells expressing the antibody fragment on the cell surface. These data indicate that antibody-based reporter genes represent a promising new platform for the development of new reporter gene and probe combinations.
Antibody-based reporter genes have several potential advantages over other combinations. For example, the pharmacokinetics of the reporter probe can easily be optimized, and probes can identify antibodies with much higher specificity, thus improving the accuracy of PET imaging. In addition, the number of antibodies that can be used as reporter genes is virtually unlimited compared with available viral or mammalian reporter genes. Antibody-based reporter genes have low immunogenicity and are better suited for imaging the expression of several genes.
Source: Society of Nuclear Medicine