Researchers explain spread of 1918 flu pandemic
MIT researchers have explained why two mutations in the H1N1 avian flu virus were critical for viral transmission in humans during the 1918 pandemic outbreak that killed at least 50 million people.
The team showed that the 1918 influenza strain developed two mutations in a surface molecule called hemagglutinin (HA), which allowed it to bind tightly to receptors in the human upper respiratory tract.
“Two mutations dramatically change the HA binding affinity to receptors found in the human upper airways,” said Ram Sasisekharan, the Underwood Prescott Professor of Biological Engineering and Health Sciences and Technology.
Sasisekharan is the senior author of a paper on the work to be published in the Feb. 18 issue of the Proceedings of the National Academy of Sciences.
In January, Sasisekharan and colleagues reported in Nature Biotechnology that flu viruses can only bind to human respiratory cells if they match the shape of sugar (or glycan) receptors found on those cells.
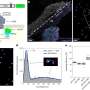
The glycan receptors found in the human respiratory tract are known as alpha 2-6 receptors, and they come in two shapes-one resembling an open umbrella, and another resembling a cone. To infect humans the MIT team found that avian flu viruses must gain the ability to bind to the umbrella-shaped alpha 2-6 receptor.
In the current study, the team discovered that two mutations in HA allow flu viruses to bind tightly or with high affinity to the umbrella-shaped glycan receptors.
“The affinity between the influenza virus HA and the glycan receptors appears to be a critical determinant for viral transmission,” said Sasisekharan.
The researchers used the 1918 influenza virus as a model system to investigate the biochemical basis for hemagglutinin binding to glycans, which leads to viral transmission. They compared the virus that caused the 1918 pandemic (known as SC18) with a strain called NY18 that differs from SC18 by only one amino acid, and also the AV18 strain, which differs from SC18 by two amino acids.
Using ferrets (which are susceptible to human flu strains), researchers had earlier found that, while SC18 transmitted efficiently between ferrets, NY18 is only slightly infectious and AV18 not at all infectious.
These earlier findings correlate with the viruses' ability to bind umbrella-shaped alpha 2-6 glycan receptors, demonstrated in the current PNAS study.
NY18, which is only slightly infectious, binds to the umbrella-shaped alpha 2-6 receptors but not as well as SC18, which is highly infectious.
AV18, which does not infect humans, does not have any affinity for the umbrella-shaped alpha 2-6 receptors and binds only to alpha 2-3 receptors.
Another strain, TX18, binds to alpha 2-6 and alpha 2-3 but is much more infectious than NY18, because it binds with high affinity to the umbrella-shaped alpha 2-6 receptors.
Researchers from the Centers for Disease Control and Prevention reported on the varying infectiousness of these strains last year, but the PNAS study is the first that explains the exact biochemical reason underlying these differences.
This new work could aid researchers in monitoring the HA mutations in the H5N1 avian flu strains currently circulating in Asia. These mutations could enable the virus to jump from birds to humans, as many epidemiologists fear will occur.
Source: Massachusetts Institute of Technology