Team discovers how brain's own tPA helps regulate blood flow to neurons
The human brain contains its own store of a powerful enzyme (and stroke drug) called tissue plasminogen activator (tPA), which appears to be a key regulator of blood flow to brain cells, a team at the Weill Cornell Medical College in New York City reports.
"We found that this natural tPA boosts blood flow to brain cells via its influence on nitric oxide synthase, which is essential to the production of nitric oxide (NO). NO is a well-known vasodilator -- a drug or chemical that widens blood vessels -- so, more NO means better blood flow to neurons as they become more active," explains study senior author Dr. Costantino Iadecola, the George C. Cotzias Distinguished Professor of Neurology and Neuroscience at Weill Cornell Medical College, and chief of the Division of Neurobiology at NewYork-Presbyterian Hospital/Weill Cornell Medical Center.
The findings have just been published in this week's online issue of Proceedings of the National Academies of Science.
Besides elucidating the role of naturally produced tPA in neuronal blood flow, the new findings could have implications for the study of stroke and Alzheimer's disease. Both conditions are associated with marked declines in natural brain levels of tPA.
TPA has become a star of sorts in cardiovascular research over the past two decades, ever since scientists discovered its remarkable ability to break up clots.
"Essentially, tPA, a powerful protease enzyme, cleaves a protein called plasminogen into plasmin, an enzyme that quickly 'eats up' clots," notes study lead author Dr. Laibaik Park, instructor in neuroscience at Weill Cornell. For that reason, doctors often administer a shot of tPA to stroke patients within minutes or hours of an attack.
But other research had also detected tPA occurring naturally in the human brain, with levels of the enzyme rising as brain cell activity increased.
"What really piqued our interest was the finding from recent studies that tPA somehow modulates the activity of a protein lying on the surface of neurons called the NMDA receptor," Dr. Iadecola explains. "This receptor serves as a gateway of communication between adjoining neurons, with glutamate being the 'currency' of exchange. Fluctuating levels of tPA seemed to influence just how much of that currency got through as brain cells became more or less active."
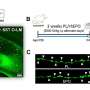
Exploring this mechanism further, Dr. Iadecola's team utilized a genetically engineered "knockout" mouse that lacked neuronal tPA. They tweaked the mouse's whiskers and watched blood flow in the area of the rodent's brain linked to whisker sensitivity.
"In the knockout mouse, blood flow in that area did not change as much upon whisker stimulation -- confirming that tPA is necessary to boosting local blood flow," Dr. Iadecola says.
But how was tPA working, exactly? The prevailing theory -- that the enzyme impacted directly on the NMDA receptor -- was quickly proven wrong. "We found that tPA was not acting as any kind of direct 'choke' on the NMDA receptor to allow more or less glutamate into the cell," says Eduardo Gallo, a graduate student in the Department of Neurology and Neuroscience, who played a key role in the study.
So, the team looked elsewhere at other rate-limiting mechanisms that might explain tPA's effects.
"One of the end-products of NMDA receptor activity is nitric oxide (NO), a powerful vasodilator," Gallo notes. "In our experiments, we discovered that tPA helps control how much NO can be made by activation of the NMDA receptor. TPA does so by boosting the ability of neuronal nitric oxide synthase (nNOS) -- an enzyme -- to produce NO. More tPA means more active nitric oxide synthase -- and more of this enzyme means more vessel-widening NO. The end result: a localized boost in blood flow to brain cells."
Questions remain, however. TPA exists outside the brain cell, but the nNOS activity and NO production goes on inside the neuron, Dr. Iadecola points out. "That means there's some kind of biochemical chain connecting external tPA to these internal mechanisms," he says. "Identifying the key players in that pathway will be a key part of our research going forward."
The new discoveries will have exciting implications for brain research, he says.
"More and more, we are realizing that alterations in the availability of blood to brain cells is crucial to stroke and post-stroke recovery, and in the debilitating loss of neuronal function that underlies Alzheimer's disease and other dementias," Dr. Iadecola says.
"It is possible that drugs or other interventions that manipulate the brain's natural supply of tPA could help preserve neurological function after stroke or Alzheimer's, or even help reverse some of the damage," he says. "Those types of treatments are still a long way off, but our new understanding how tPA works to keep neurons healthy and active is a crucial first step in that research."
Source: New York- Presbyterian Hospital