Researchers find new way to block destructive rush of immune cells
Researchers have found a way to selectively block the ability of white blood cells to “crawl” toward the sites of injury and infection when such mobility drives disease, according to a study published today in The Journal of Experimental Medicine. The results suggest a new treatment approach for autoimmune diseases like rheumatoid arthritis, lupus and multiple sclerosis, and for conditions made worse by misplaced inflammation, like atherosclerosis, stroke and transplant rejection, researchers said.
Where a single-celled amoeba moves to find food, human cells migrate as part of complex bodily functions like immunity. Disease-fighting cells for instance move toward bacteria and cells infected with viruses, which they target for destruction. Unfortunately, the same cells can mistakenly attack the body’s own cells or drive inflammation too far, worsening the problem they rushed in to solve.
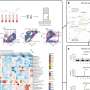
A team of researchers at the University of Rochester Medical Center has been studying proteins called integrins that enable T cells, a major subset of immune cells, to migrate. The integrin-related mechanisms described for the first time in the current paper suggest a way to shut down only those T cells currently in the act of disease-related migration, while leaving in place reserves needed in the likely event that another infection occurs during treatment. Making the mechanistic discoveries possible was a successful effort by the team to capture on video the first detailed images of fast-migrating T cells and the behavior of key proteins related to migration, which had been tagged with fluorescence. Twelve videos of T cells, and their key migration proteins, in action are part of the publication and are available online.
“There are many cases where it would be incredibly useful to precisely block integrin activation, and thus T cell migration,” said Minsoo Kim, Ph.D., assistant professor of Microbiology and Immunology within the David H. Smith Center for Vaccine Biology and Immunology at the Medical Center, and lead author of the article. “Good examples include when our immune system attacks our own cells, or rejects a lifesaving transplant or clogs our blood vessels by mistake. The problem is that past, system-wide attempts that block all integrin activation, like the multiple sclerosis drug Tysabri, shut down not only unwanted inflammation in one locale, but also vital immune defenses elsewhere, leaving patients vulnerable to infection.”
The Great Migration
Two mechanisms make cell migration, or programmed directional movement, possible. The first, called chemotaxis, tells the cell which direction to move in. Cell surface proteins sense and follow chemicals and molecules they are attracted to toward wherever those attractants are most concentrated. T cells, named after the thymus (T) where they mature, move toward the byproducts of bacteria and viruses.
The second migratory mechanism is propulsion. In between infections and injuries, inactive T cells ride along with the bloodstream. T cells “realize” when they pass by part of a blood vessel wall close to the site of an injury or infection. Integrins on their surfaces unfold and grab onto key proteins on the surface of blood vessel wall cells (e.g. ICAM), resisting the surrounding blood flow. The T cells then pass through the vessel wall, and once outside the bloodstream, crawl along the tissue scaffolding toward the site of injury.
In a T cell at rest, integrins are distributed evenly over the entire surface of the T cell. When the cell gets ready to move, however, activated integrins cluster on the leading edge of the cell in the direction the cell wants to move in. They bind to their counterpart adhesion proteins like ICAM on the surface that the T cell is moving across. The T cell then contracts using its cell skeleton to pull itself over the leading edge integrins. Finally, the integrins on the trailing edge of the cell let go. Without precise changes that enable the front end to gain traction, and the tail to let go, the cell cannot migrate.
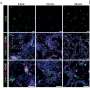
Kim’s team found that a subset of integrins, including lymphocyte function–associated antigen-1 (LFA-1), control whether or not the tail end of the T cell can “let go” (de- adhesion). Data revealed for the first time that a protein called non-muscle myosin heavy chain-IIA (MyH9) is recruited to LFA-1 at the trailing end of migrating T lymphocytes. Experiments that interfered with the association between MyH9 and the LFA-1 integrin were found to prevent the trailing edge of the crawling T cell from letting go, dramatically reducing the ability of T cells to move. Myosins are motor proteins that expend energy to enable cell skeletons to contract. That contraction creates force that is used in many cases to move muscle fibers, but in the case of MyH9, to rip the trailing end of a migrating T cell foot away from the surface it is migrating across by breaking integrin-ICAM bonds. The results provide the first evidentiary support of the longstanding theory that cell skeleton contractile force is used to drive T cell migration, with MyH9 as the mechanical link. Captured images show fluorescently tagged actin (which partners with LFA-1 to grip the surface) gathering at the front end of the cell, and fluorescently tagged MyH9 gathering at the tail end in cycles, each time the cell takes a “step.”
The study was a joint effort by the Department of Surgery at Rhode Island Hospital, Brown Medical School, the Department of Physics at Brown University, the CBR Institute for Biomedical Research at Harvard Medical School and the departments of Chemical Engineering, Biomedical Engineering and Department of Microbiology and Immunology at the University of Rochester. The project was supported by the American Heart Association, the Rhode Island Foundation, the National Institutes of Health, the National Science Foundation and the Brown University Seed Grant.
In the next phase, the team will seek to develop better-targeted, anti-integrin therapies, with MyH9 among the rational targets for new classes of drugs. Toward that end, experiments currently underway are designed to determine which molecules regulate MyH9 activity during T cell migration.
“Initial clinical studies on T cell migration focused on overall blocking of migration, but general inhibition is a blunt tool,” said Tim Mosmann, Ph.D., director of the David H. Smith Center for Vaccine Biology and Immunology. “As studies such as Dr. Kim’s help us to understand the process more precisely, we should be able to design much more precise methods to block migration in the selected circumstances that cause problems, without crippling the essential immune responses to infections.”
Source: University of Rochester Medical Center